Final ID: Or112
Macrophage-Targeted Oxygen-Generating Theranostic Photoactivation Facilitates Inflammation Resolution and Stabilization of Hypoxic Atherosclerotic Plaques
Abstract Body: Introduction: Atherosclerosis is a chronic inflammatory disease driven by the dense infiltration of immune cells, particularly M1-polarized macrophages, within arterial plaques. Photoactivation presents a promising theranostic strategy by generating reactive oxygen species to eliminate inflammatory cells and promote healing. However, as lesions advance, the localized hypoxic microenvironment stabilizes hypoxia-inducible factor-1α (HIF-1α), which not only exacerbates pro-inflammatory signaling and M1 polarization but also severely limits the efficacy of oxygen-dependent treatment. We hypothesized that macrophage-targeted, oxygen-generating theranostic photoactivation could alleviate hypoxia, driving inflammation resolution and stabilizing hypoxic atherosclerotic plaques.
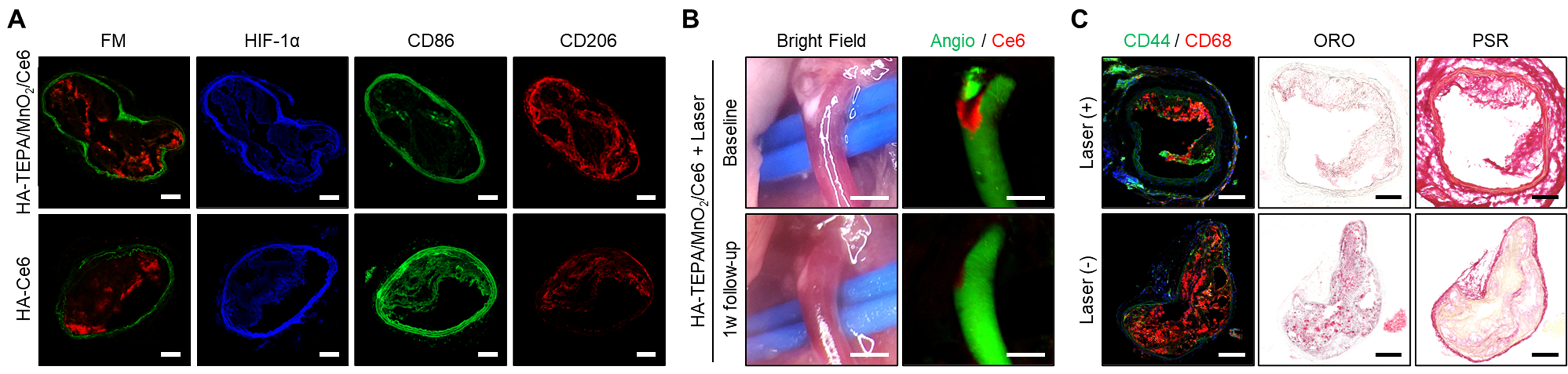
Method and results: We synthesized a macrophage-targeted, oxygen-generating, photoactivatable agent comprising hyaluronic acid for CD44 ligand, manganese dioxide (MnO2) for in situ oxygen generation via H2O2 decomposition, and chlorin e6 (Ce6) for near-infrared fluorescence (NIRF) imaging and phototherapy (HA-TEPA/MnO2/Ce6). In hypoxic atherosclerotic murine models, we intravenously administered a multifunctional agent at a Ce6-equivalent dose of 3 mg/kg. In vivo NIRF imaging confirmed that the agents selectively accumulated in CD68+ macrophage-rich regions within the plaques. Notably, the MnO2-mediated oxygen generation significantly attenuated HIF-1α expression and triggered a phenotypic shift from M1 to M2 macrophages (p < 0.01). Serial intravital imaging revealed that oxygenation-enhanced photoactivation exhibited a remarkable reduction in inflammatory NIRF signals within plaques at 1-week post-laser irradiation. Histologically, oxygen-generating photoactivation induced inflammation resolution and stabilization (p < 0.01). Together, these findings demonstrated the synergistic effect of the MnO2-mediated mechanism and oxygenation-enhanced phototherapy enabled effective plaque regression and stabilization.
Conclusion: Macrophage-targeted oxygen-generating theranostic photoactivation effectively reverses the hypoxic-inflammatory milieu of advanced plaques. By alleviating hypoxia and driving M1-to-M2 polarization, this agent enables oxygen-augmented photoactivation that not only resolves inflammation but also stabilizes hypoxic atherosclerotic plaques. This study highlights a transformative theranostic approach for personalized management of coronary artery disease.
Method and results: We synthesized a macrophage-targeted, oxygen-generating, photoactivatable agent comprising hyaluronic acid for CD44 ligand, manganese dioxide (MnO2) for in situ oxygen generation via H2O2 decomposition, and chlorin e6 (Ce6) for near-infrared fluorescence (NIRF) imaging and phototherapy (HA-TEPA/MnO2/Ce6). In hypoxic atherosclerotic murine models, we intravenously administered a multifunctional agent at a Ce6-equivalent dose of 3 mg/kg. In vivo NIRF imaging confirmed that the agents selectively accumulated in CD68+ macrophage-rich regions within the plaques. Notably, the MnO2-mediated oxygen generation significantly attenuated HIF-1α expression and triggered a phenotypic shift from M1 to M2 macrophages (p < 0.01). Serial intravital imaging revealed that oxygenation-enhanced photoactivation exhibited a remarkable reduction in inflammatory NIRF signals within plaques at 1-week post-laser irradiation. Histologically, oxygen-generating photoactivation induced inflammation resolution and stabilization (p < 0.01). Together, these findings demonstrated the synergistic effect of the MnO2-mediated mechanism and oxygenation-enhanced phototherapy enabled effective plaque regression and stabilization.
Conclusion: Macrophage-targeted oxygen-generating theranostic photoactivation effectively reverses the hypoxic-inflammatory milieu of advanced plaques. By alleviating hypoxia and driving M1-to-M2 polarization, this agent enables oxygen-augmented photoactivation that not only resolves inflammation but also stabilizes hypoxic atherosclerotic plaques. This study highlights a transformative theranostic approach for personalized management of coronary artery disease.
More abstracts on this topic:
Association of TTR-Targeted Therapies with Cardiovascular Outcomes in Transthyretin Amyloid Cardiomyopathy: A Real-World Analysis
Shahid Abdulla, Arora Pankaj, Pampana Akhil, Gaonkar Mokshad, Patel Nirav, Bal Harshvir, Nayak Amrita, Vekariya Nehal, Shetty Naman, Arora Garima
A Case Report: Outpatient Diagnosis of Venous Stent Migration - Avoiding Catastrophic OutcomesBasnyat Anouksha, Pamganamamula Madhu, Naidu Raja, Pamganamamula Teja, Manchiraju Srinidhi, Gaddam Srilakshmi, Panganamamula Lalitha