Final ID: Fri005
The Histone Acetyltransferase MOF Regulates Macrophage Inflammation During Abdominal Aortic Aneurysm Development
Abstract Body: Objective: Abdominal aortic aneurysms (AAAs) are a life-threatening disease for which there is a lack of effective therapy preventing rupture. During AAA formation, pathological vascular remodeling is driven by macrophage infiltration, and the mechanisms regulating macrophage-mediated inflammation remain undefined. Recent evidence suggests that MOF, a histone acetyltransferase that acetylates histone 4 lysine 16 (H4K16), has been shown to regulate inflammatory gene transcription, however its role in macrophage inflammation and cardiovascular disease remains to be defined. The objective of this study was to determine the role of MOF in regulating macrophage inflammation and AAA development.
Methods: Single-cell RNA sequencing was performed on human AAA and non-aneurysmal control aortic tissue to analyze MOF expression in monocyte/macrophage populations (n=14). Two murine AAA models were utilized, including angiotensin II (AngII) infusion and topical elastase treatment. To investigate the role of MOF in AAA formation, mice underwent pharmacologic inhibition with MG-149 (1mg/kg) or macrophage-specific genetic deletion of MOF (Mof−/−MΦ). Macrophage inflammatory gene expression was analyzed by qPCR, ELISA, and flow cytometry. Chromatin immunoprecipitation was conducted to assess levels of H4K16ac in human and murine AAA macrophages.
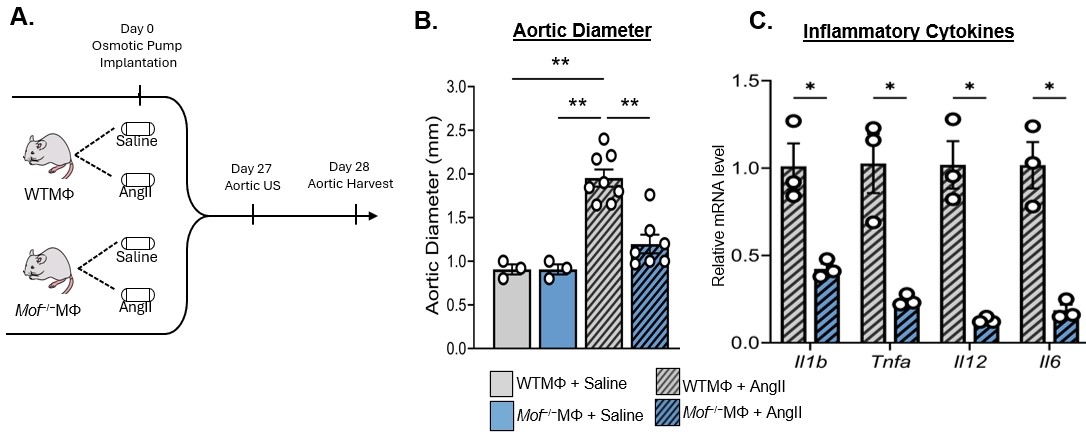
Results: Single-cell transcriptomic analysis demonstrated that MOF expression was increased in monocytes/macrophages from human AAA tissue compared to non-aneurysmal controls. MOF expression was similarly elevated in macrophages from both AngII- and elastase-induced murine AAAs. Increased MOF expression was associated with increased H4K16ac deposition on NF-κB–regulated inflammatory gene promoters, including Il1b and Tnfa, resulting in enhanced inflammatory gene transcription and altered macrophage phenotype. Pharmacologic inhibition or macrophage-specific genetic deletion of MOF (Mof−/−MΦ) significantly reduced H4K16ac levels within macrophages and promoted an anti-inflammatory phenotype (Figure 1A-C). Additionally, MOF inhibition reduced AAA formation, preserved aortic wall architecture, decreased elastin fragmentation, and attenuated macrophage-mediated inflammation.
Conclusions: MOF regulates macrophage inflammation during AAA development through epigenetic control of NF-κB–mediated inflammatory gene transcription. Targeting the MOF pathway may provide a therapeutic strategy to prevent AAA progression.
Methods: Single-cell RNA sequencing was performed on human AAA and non-aneurysmal control aortic tissue to analyze MOF expression in monocyte/macrophage populations (n=14). Two murine AAA models were utilized, including angiotensin II (AngII) infusion and topical elastase treatment. To investigate the role of MOF in AAA formation, mice underwent pharmacologic inhibition with MG-149 (1mg/kg) or macrophage-specific genetic deletion of MOF (Mof−/−MΦ). Macrophage inflammatory gene expression was analyzed by qPCR, ELISA, and flow cytometry. Chromatin immunoprecipitation was conducted to assess levels of H4K16ac in human and murine AAA macrophages.
Results: Single-cell transcriptomic analysis demonstrated that MOF expression was increased in monocytes/macrophages from human AAA tissue compared to non-aneurysmal controls. MOF expression was similarly elevated in macrophages from both AngII- and elastase-induced murine AAAs. Increased MOF expression was associated with increased H4K16ac deposition on NF-κB–regulated inflammatory gene promoters, including Il1b and Tnfa, resulting in enhanced inflammatory gene transcription and altered macrophage phenotype. Pharmacologic inhibition or macrophage-specific genetic deletion of MOF (Mof−/−MΦ) significantly reduced H4K16ac levels within macrophages and promoted an anti-inflammatory phenotype (Figure 1A-C). Additionally, MOF inhibition reduced AAA formation, preserved aortic wall architecture, decreased elastin fragmentation, and attenuated macrophage-mediated inflammation.
Conclusions: MOF regulates macrophage inflammation during AAA development through epigenetic control of NF-κB–mediated inflammatory gene transcription. Targeting the MOF pathway may provide a therapeutic strategy to prevent AAA progression.
More abstracts on this topic:
Aorta to Ilio-femoral Bypass During Total Arch Replacement with Frozen Elephant Trunk Facilitates Second-Stage Thoracoabdominal Aortic Repair with Normothermic Iliac Perfusion
Zhao Ruojin, Liu Yanxiang, Wang Luchen, Sun Xiaogang
Biaxial Mechanics of Murine Abdominal Aortic Aneurysm Tissue to Model Human DiseaseWahidi Ryan, Otani Brendan, Elizondo Benedetto Santiago, Arif Batool, Braasch Maxwell, Bersi Matthew, Zayed Mohamed