Final ID: WE524
Metabolomic Analysis in Three US Cohorts With 40 Years of Follow-Up Identifies Metabolomic Profiles Reflecting Metabolic States Associated with Long-Term Obesity Trajectory and Its Related Chronic Disease Risk.
Abstract Body: Background
Obesity, a leading risk factor for coronary artery disease (CHD) and other chronic diseases, is a multifactorial condition with heterogenous etiologies and comorbidity profiles.
Hypothesis
Circulating metabolome can capture metabolic states associated with obesity trajectory and inter-person variation in obesity-related disease risk.
Methods
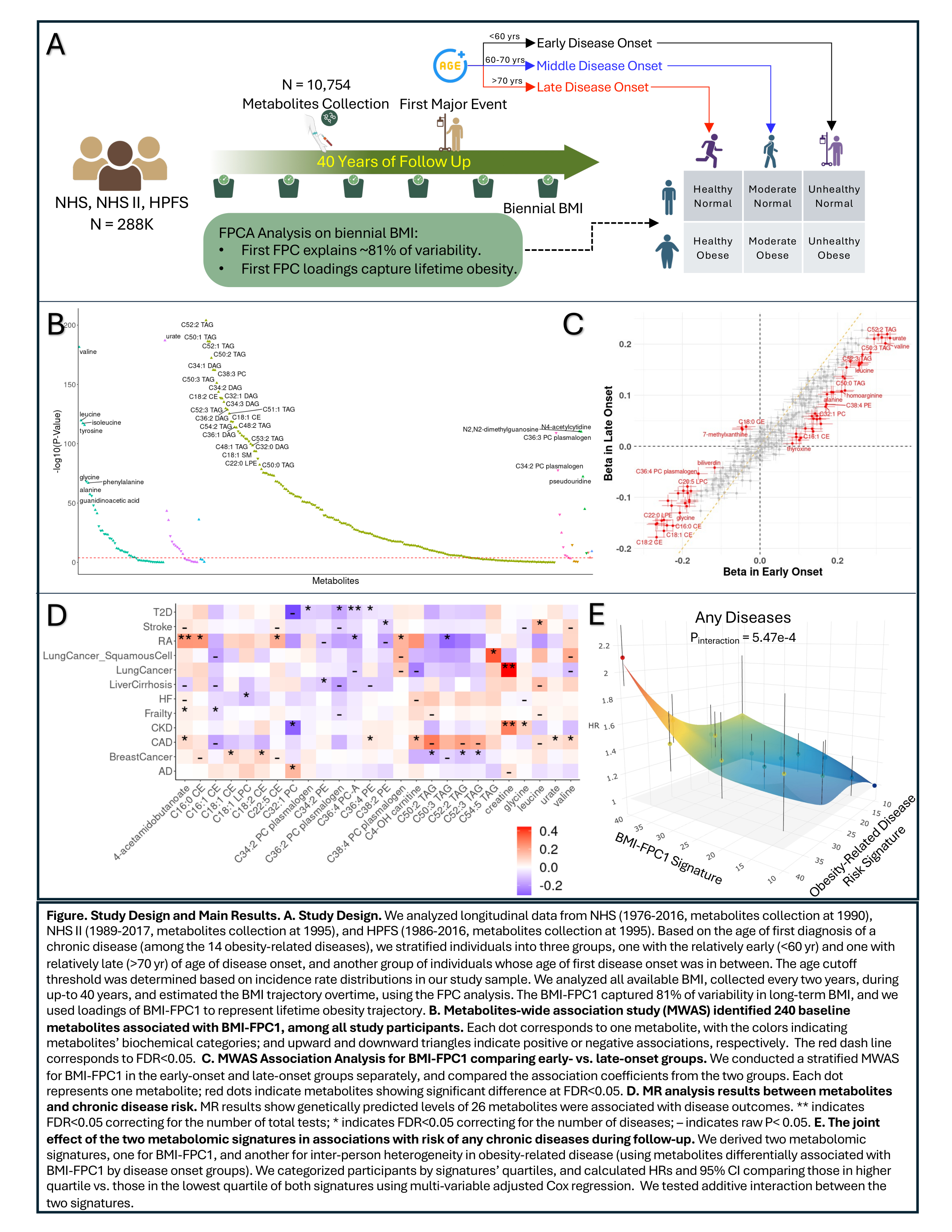
We analyzed up to 40-yr of longitudinal data of 10754 participants from the Nurses’ Health Studies and Health Professionals Follow-Up Study. Baseline plasma levels of 288 metabolites were profiled using LC-MS. Body mass index (BMI) was collected biennially, and its trajectory was estimated using function principal component (FPC) analysis. We categorize participants as having early- (<60y) or late-onset (>70y) obesity-related diseases based on age of first onset of 14 chronic diseases (Fig A). Linear regression was used to examine metabolites-BMI trajectory associations; elastic net regression to derive metabolomic signatures for BMI trajectory; Cox model to examine association with disease risk; and Mendelian randomization (MR) analysis to infer potential causal relationships.
Results
The FPC1 of BMI trajectory accounted 81% of variation. We identified extensive associations between baseline metabolites with BMI-FPC1 (240 at FDR<0.05; Fig B). Further stratified analysis identified 63 metabolites, including glycine, alanine and C52:2 TAG, showing stronger associations with BMI-FPC1 among participants with early-onset vs late-onset of obesity-related diseases (Fig C). In MR analysis, genetically predicted levels of 26 metabolites were associated with at least one of these diseases (e.g., C4-OH carnitine with CHD; Fig D).
We identified a metabolomic signature for BMI-FPC1, which was associated with risk of any chronic disease in multivariable-adjusted analysis (HR=1.99, p=4e-47). A second metabolomic signature, derived from the 63 metabolites differentially associated with BMI-FPC1 between two disease groups, was associated with disease risk after adjusting for the BMI-FPC1 signature (HR=1.2, p=5e-10). The two signatures showed an additive effect (p-interaction=6e-4), with participants in the highest vs. lowest quartiles of both signatures having a 11.3-fold higher disease risk (p=3e-50; Fig E).
Conclusions
We identified metabolomic profiles reflecting metabolic states related to long-term BMI trajectory and inter-individual variation in obesity-related disease risk, which may facilitate personalized intervention.
Obesity, a leading risk factor for coronary artery disease (CHD) and other chronic diseases, is a multifactorial condition with heterogenous etiologies and comorbidity profiles.
Hypothesis
Circulating metabolome can capture metabolic states associated with obesity trajectory and inter-person variation in obesity-related disease risk.
Methods
We analyzed up to 40-yr of longitudinal data of 10754 participants from the Nurses’ Health Studies and Health Professionals Follow-Up Study. Baseline plasma levels of 288 metabolites were profiled using LC-MS. Body mass index (BMI) was collected biennially, and its trajectory was estimated using function principal component (FPC) analysis. We categorize participants as having early- (<60y) or late-onset (>70y) obesity-related diseases based on age of first onset of 14 chronic diseases (Fig A). Linear regression was used to examine metabolites-BMI trajectory associations; elastic net regression to derive metabolomic signatures for BMI trajectory; Cox model to examine association with disease risk; and Mendelian randomization (MR) analysis to infer potential causal relationships.
Results
The FPC1 of BMI trajectory accounted 81% of variation. We identified extensive associations between baseline metabolites with BMI-FPC1 (240 at FDR<0.05; Fig B). Further stratified analysis identified 63 metabolites, including glycine, alanine and C52:2 TAG, showing stronger associations with BMI-FPC1 among participants with early-onset vs late-onset of obesity-related diseases (Fig C). In MR analysis, genetically predicted levels of 26 metabolites were associated with at least one of these diseases (e.g., C4-OH carnitine with CHD; Fig D).
We identified a metabolomic signature for BMI-FPC1, which was associated with risk of any chronic disease in multivariable-adjusted analysis (HR=1.99, p=4e-47). A second metabolomic signature, derived from the 63 metabolites differentially associated with BMI-FPC1 between two disease groups, was associated with disease risk after adjusting for the BMI-FPC1 signature (HR=1.2, p=5e-10). The two signatures showed an additive effect (p-interaction=6e-4), with participants in the highest vs. lowest quartiles of both signatures having a 11.3-fold higher disease risk (p=3e-50; Fig E).
Conclusions
We identified metabolomic profiles reflecting metabolic states related to long-term BMI trajectory and inter-individual variation in obesity-related disease risk, which may facilitate personalized intervention.
More abstracts on this topic:
A Case of Dilated Cardiomyopathy and Systemic Thromboembolism in a Young Patient on Testosterone Replacement Therapy
Sabri Muhammad, Ijaz Naila, Nadeem Ramsha, Checchio Lucy, Riaz Faiza
A First-in-Class EV-miRNA Diagnostic System for Early Identification of IVIG-Resistant Kawasaki DiseaseNakaoka Hideyuki, Hirono Keiichi, Hara Akane, Tsuboi Kaori, Ibuki Keijiro, Ozawa Sayaka, Ichida Fukiko