Final ID: MP2630
From Gut to Lung: Lipoxygenase Dependent Oxylipin Amplification Drives 15-HETE Induced Pulmonary Hypertension in C57BL6/J Mice
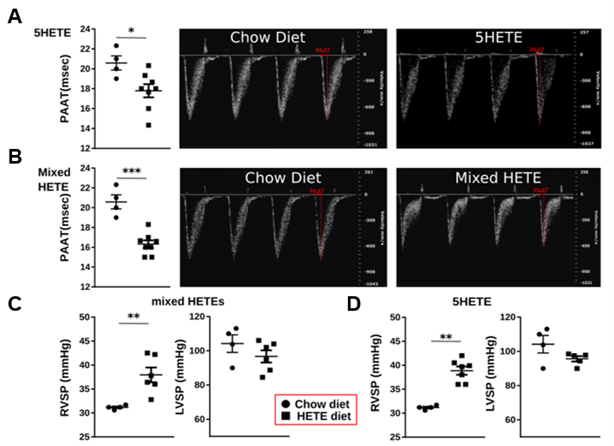
Abstract Body (Do not enter title and authors here): Background: Pulmonary arterial hypertension (PAH) is a progressive and fatal disease characterized by pulmonary vascular remodeling, increased right ventricular pressure, and eventual heart failure. Elevated levels of oxidized lipid metabolites especially hydroxyeicosatetraenoic acids (HETEs) have been observed in both human patients and animal models, implicating these lipids in the disease’s pathogenesis. We previously demonstrated that dietary supplementation with 15-HETE for 3 weeks is sufficient to induce pulmonary hypertension (PH) in C57BL6/J. However, the molecular mechanisms remain poorly understood. During our investigations, we discovered that dietary 15-HETE increases systemic oxylipin levels of not only 15HETE but also other HETEs (including 5-, 12-), like PAH patients. We therefore hypothesized that 15-HETE promotes PH through an initial lipoxygenase (LOX)-mediated oxylipin amplification, which in turn drives metabolic and immune alterations in the gut and contribute to progression of PH. Methods: PH development was assessed by pulmonary arterial acceleration time (PAAT) and right ventricular systolic pressure (RVSP). LOX involvement was tested by blocking total LOX activity, treating 12/15-LOX-deficient mice with a 5-LOX inhibitor (zileuton). Plasma lipid profiles were analyzed mass spectrometry. Cytokine levels were measured by multiplex immunoassay. Flow cytometry was used to evaluate gut immune populations. Results: 15-HETE alone, 5-HETE alone or combined 5-HETE and 12-HETE supplementation induced PH, reflected by significantly decreased PAAT and elevated RVSP. Lipidomics analysis revealed increased oxilipins in 15-HETE treated mice. Elevation in plasma inflammatory cytokines (including eotaxin, G-CSF, IL-15, and MCP-1) and shifts in gut immune cells (increased Type 1 macrophages and decreased neutrophils) were observed in 15-HETE treated mice, suggesting an inflammatory response initiated in the gut. Finally, 12/15-LOX deficiency combined with zileuton treatment not only reduced intestinal 5-, 12-, 15-HETE levels but also prevented 15-HETE induced PH suggesting that 15-HETE mediated oxylipin amplification is causal in the development of PH in mice treated with 15-HETE. Conclusions: Our findings suggest that the gut appears to play a previously underappreciated role in the development of PH, highlighting the potential of targeting LOX pathways and gut–lung interactions in the treatment of PAH.
More abstracts on this topic:
Transcatheter Mitral Edge-to-Edge Repair versus Surgical Mitral Valve Repair in Elderly Patients with Heart Failure: A Propensity Matched Analysis
Shehzad Mustafa, Shehzad Dawood, Yousafzai Osman, Ahmad Muhammad, Chaudhry Hammad, Vandyck-acquah Marian
Abdominal Aortic Perivascular Adipose Tissue Lipolysis Is Activated In Hypertensive Dahl SS Rats Fed a High-Fat DietChirivi Miguel, Rendon Javier, Lauver Adam, Fink Gregory, Watts Stephanie, Contreras Andres