Final ID: Mo2111
VICTORION-INCEPTION: Consistent Low-density Lipoprotein Cholesterol Lowering With Inclisiran Following Acute Coronary Syndrome Independent of Index Lipid-lowering Therapy
Abstract Body (Do not enter title and authors here): Background: In the VICTORION-INCEPTION trial (NCT04873934), participants with recent acute coronary syndrome (ACS) randomized to receive inclisiran + usual care had greater low-density lipoprotein cholesterol (LDL-C) reductions and goal attainment than those randomized to usual care alone.
Research question: Given the variability in utilization of lipid-lowering therapy (LLT) at the time of index ACS in VICTORION-INCEPTION, we sought to assess if LLT utilization at time of index ACS event impacted LDL-C goal attainment and LDL-C lowering with inclisiran.
Methods: Eligible participants were screened within 5 weeks of discharge from hospitalization for ACS with LDL-C ≥70 mg/dL (or non–high-density lipoprotein cholesterol ≥100 mg/dL) and were receiving statin therapy or had statin intolerance. Participants were randomized 1:1 to inclisiran (inclisiran sodium 300 mg [284 mg inclisiran equivalent] on Days 0, 90, and 270) + usual care or to usual care alone. We present analyses of LDL-C <70 mg/dL and <55 mg/dL goal attainment and percentage change from baseline in LDL-C by index LLT utilization (captured in the case report form as LLT used prior to index ACS event).
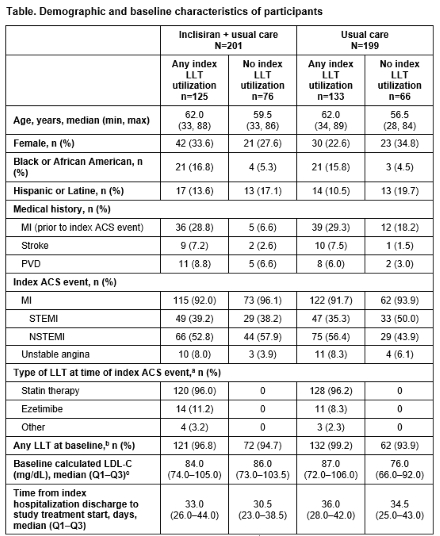
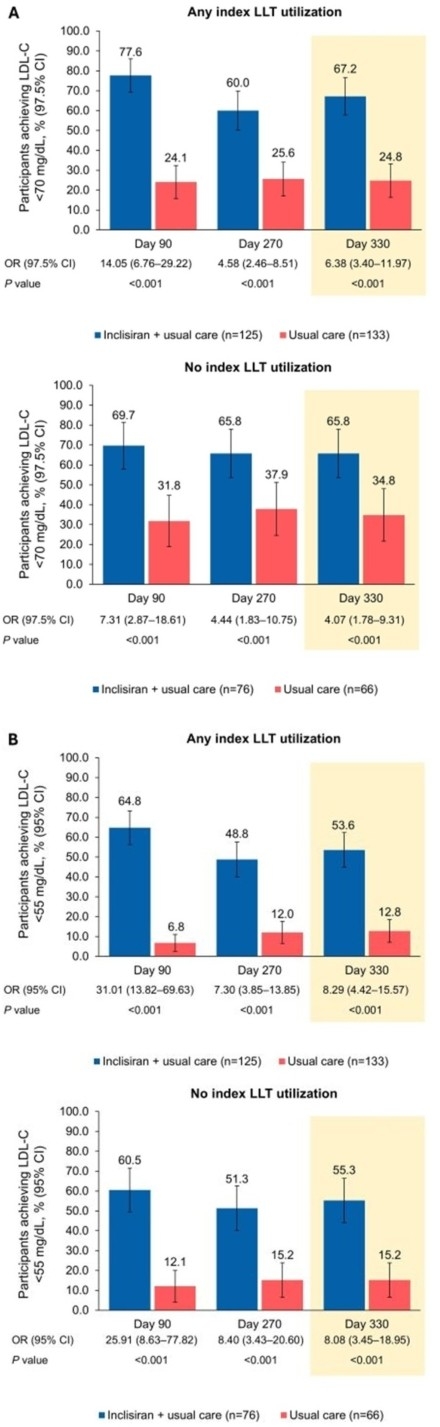
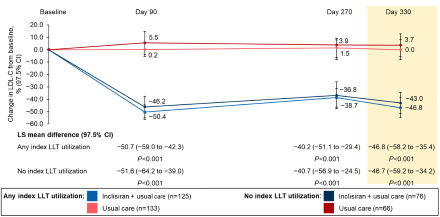
Results: Of 400 randomized participants, 64.5% were receiving index LLT (inclisiran + usual care: 62.2%; usual care: 66.8%) increasing to 96.8% of participants at the time of study treatment start (inclisiran + usual care: 96.0%; usual care: 97.5%; Table). Participants receiving index LLT had higher median age and higher rates of prior myocardial infarction. Irrespective of index LLT utilization, significantly more participants in the inclisiran + usual care arm, vs usual care, achieved LDLC <70 mg/dL from Day 90 through to Day 330 (LLT: ≥60.0% vs ≤25.6%; no LLT: ≥65.8% vs ≤37.9%; P<0.001 at all timepoints) and <55 mg/dL (LLT: ≥48.8% vs ≤12.8%; no LLT: ≥51.3% vs ≤15.2%; P<0.001 at all timepoints) (Figure 1). Similarly, LDLleast C least squares mean percentage change from baseline at Day 90 was significantly greater with inclisiran + usual care vs usual care (LLT: −50.4% vs 0.2%; no LLT: −46.2% vs 5.5%; both P<0.001) and was sustained to Day 330 (Figure 2).
Conclusion: Irrespective of index LLT utilization, in VICTORION-INCEPTION, treatment with inclisiran + usual care resulted in rapid and sustained LDL-C goal attainment and LDL-C lowering.
Research question: Given the variability in utilization of lipid-lowering therapy (LLT) at the time of index ACS in VICTORION-INCEPTION, we sought to assess if LLT utilization at time of index ACS event impacted LDL-C goal attainment and LDL-C lowering with inclisiran.
Methods: Eligible participants were screened within 5 weeks of discharge from hospitalization for ACS with LDL-C ≥70 mg/dL (or non–high-density lipoprotein cholesterol ≥100 mg/dL) and were receiving statin therapy or had statin intolerance. Participants were randomized 1:1 to inclisiran (inclisiran sodium 300 mg [284 mg inclisiran equivalent] on Days 0, 90, and 270) + usual care or to usual care alone. We present analyses of LDL-C <70 mg/dL and <55 mg/dL goal attainment and percentage change from baseline in LDL-C by index LLT utilization (captured in the case report form as LLT used prior to index ACS event).
Results: Of 400 randomized participants, 64.5% were receiving index LLT (inclisiran + usual care: 62.2%; usual care: 66.8%) increasing to 96.8% of participants at the time of study treatment start (inclisiran + usual care: 96.0%; usual care: 97.5%; Table). Participants receiving index LLT had higher median age and higher rates of prior myocardial infarction. Irrespective of index LLT utilization, significantly more participants in the inclisiran + usual care arm, vs usual care, achieved LDLC <70 mg/dL from Day 90 through to Day 330 (LLT: ≥60.0% vs ≤25.6%; no LLT: ≥65.8% vs ≤37.9%; P<0.001 at all timepoints) and <55 mg/dL (LLT: ≥48.8% vs ≤12.8%; no LLT: ≥51.3% vs ≤15.2%; P<0.001 at all timepoints) (Figure 1). Similarly, LDLleast C least squares mean percentage change from baseline at Day 90 was significantly greater with inclisiran + usual care vs usual care (LLT: −50.4% vs 0.2%; no LLT: −46.2% vs 5.5%; both P<0.001) and was sustained to Day 330 (Figure 2).
Conclusion: Irrespective of index LLT utilization, in VICTORION-INCEPTION, treatment with inclisiran + usual care resulted in rapid and sustained LDL-C goal attainment and LDL-C lowering.
More abstracts on this topic:
Access to Lipid-Lowering Therapies is Limited by Payer Coverage Restrictions and High Out-of-Pocket Costs on Medicare Prescription Drug Plans
Young Grant, Bansal Kannu, Riello Ralph, Faridi Kamil, Clark Katherine, Desai Nihar
Adding a polygenic risk score to the PREVENT clinical risk tool significantly improves cardiovascular risk predictionEuesden Jack, Absher Devin, Iribarren Carlos, Riveros-mckay Fernando, Rana Jamal, Rowell Sarah, Neogi Arpita, Harrison Seamus, Weale Michael, Donnelly Peter