Final ID: Or130
The Repurposing of FDA-Approved Hypoxia-Inducible Factor 1 Agonists is an Alternative Therapeutic Option for Deep Vein Thrombosis
Abstract Body: OBJECTIVE. Deep vein thrombosis (DVT) is a debilitating and costly condition treated by anti-coagulation, thrombectomy, or thrombolysis. These treatments prevent thrombus propagation, but increase the risk of bleeding, infection, and re-thrombosis, and fail to promote the natural thrombus resolution. Thrombus resolution is mediated by the hypoxia-inducible factor 1 (HIF1)-vascular endothelial growth factor (VEGF) pathway. Our aim was to determine whether FDA-approved HIF1 agonists can be repurposed to safely treat murine DVT.
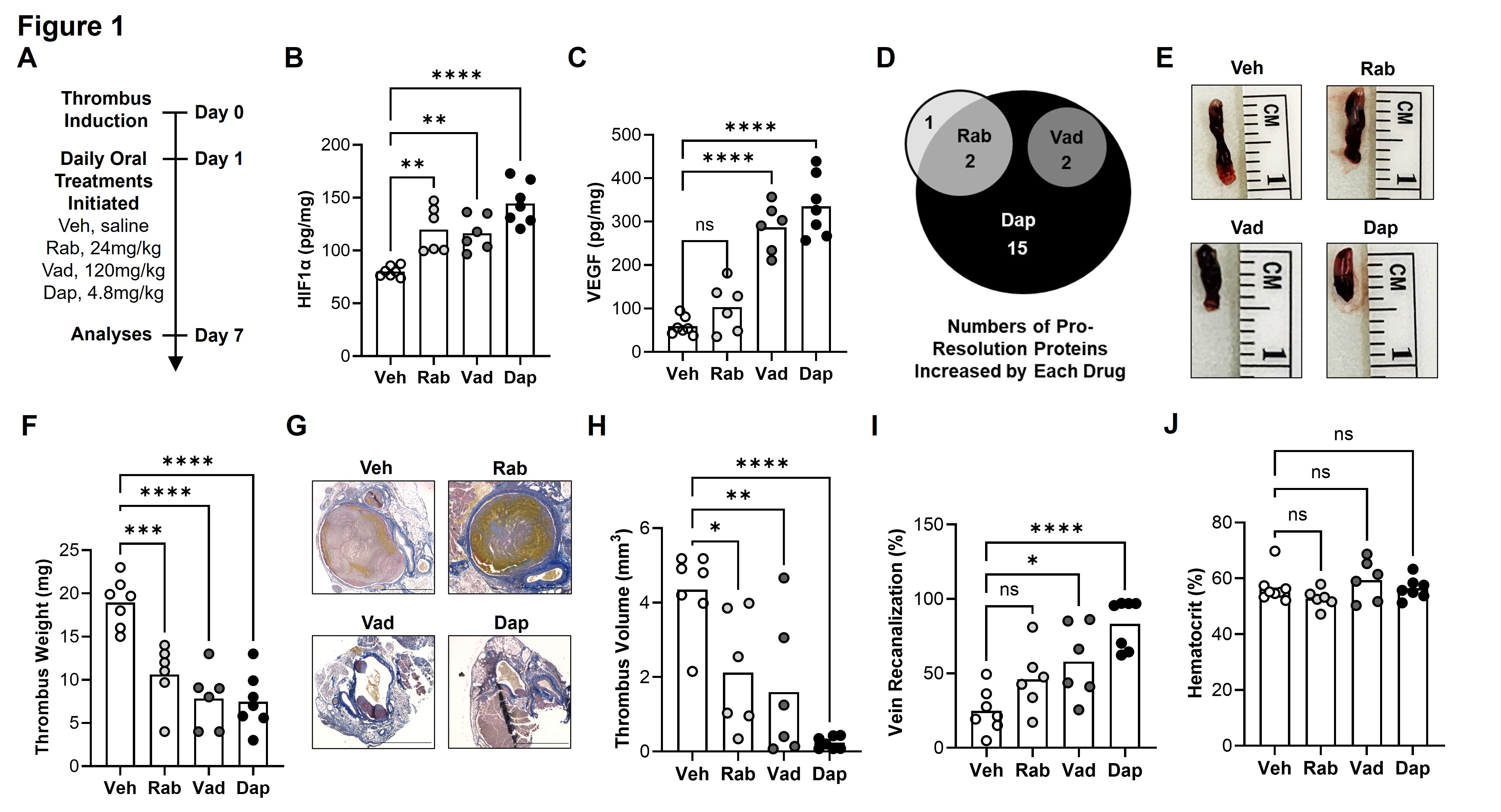
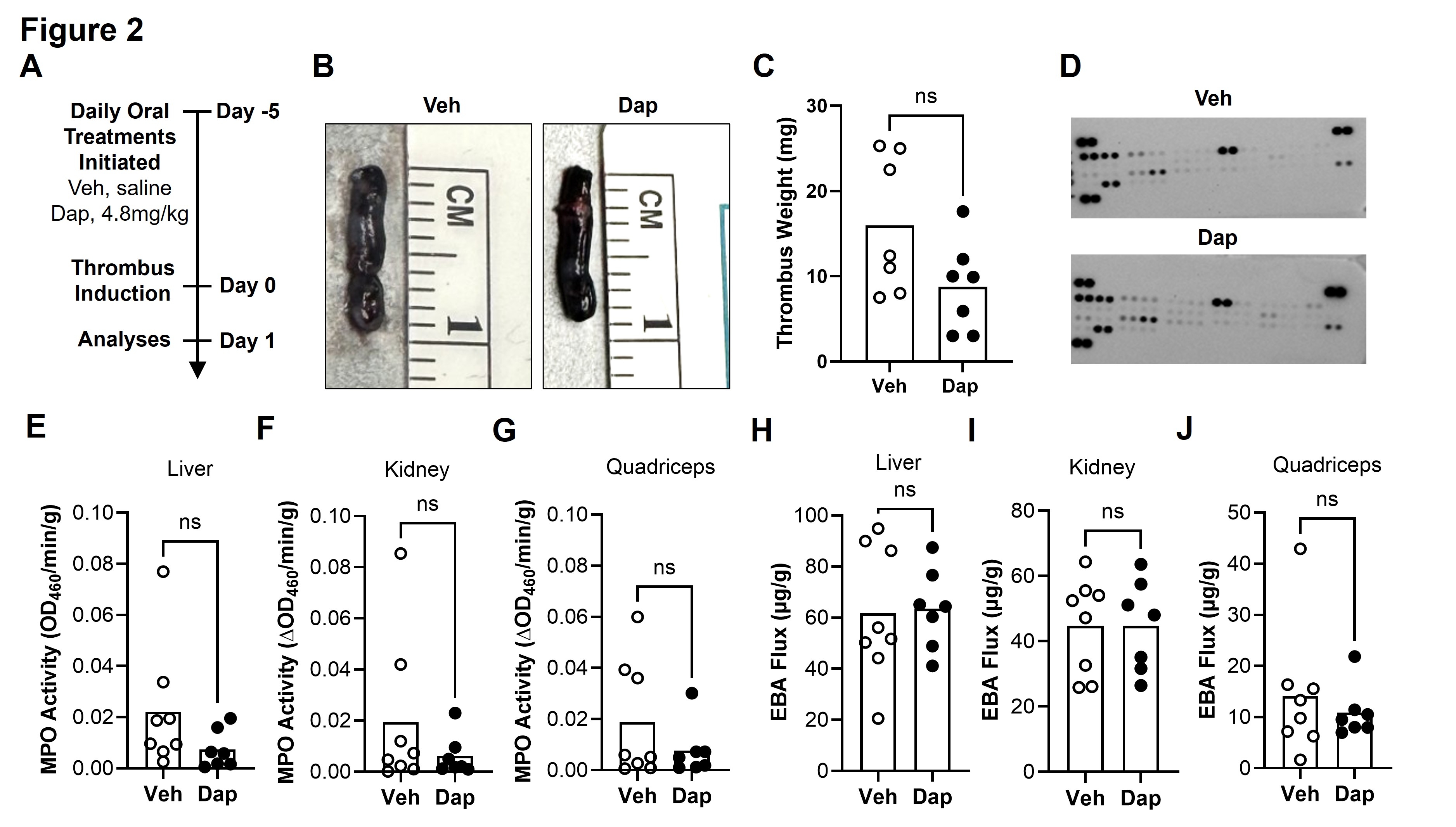
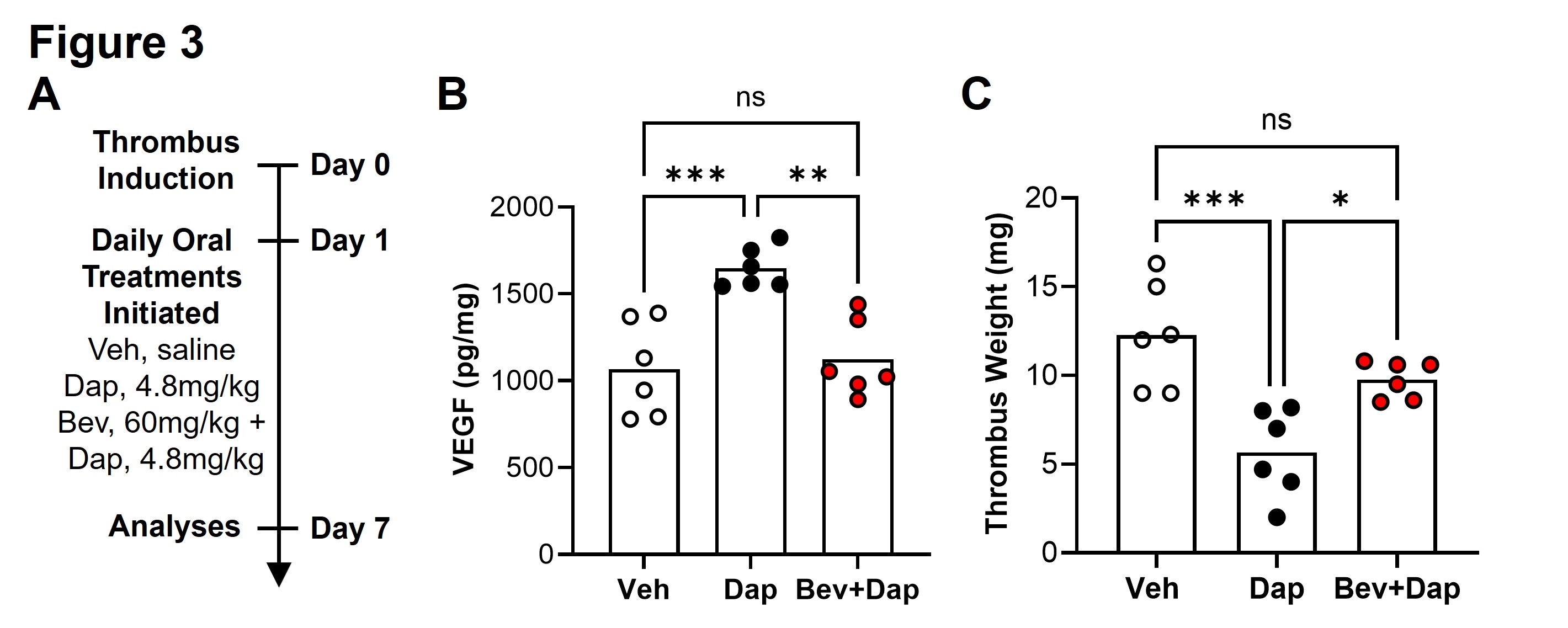
APPROACH AND RESULTS. DVT was induced by inferior vena cava stenosis in male and female C57BL/6J mice (3-4m old). Mice were treated with oral vehicle or a clinically equivalent regimen of Rabeprazole (a novel HIF1 agonist), Vadadustat or Daprodustat (known HIF1 agonists). To assess efficacy, treatments were started at day 1 post-thrombus induction, when the thrombus is fully formed (Fig. 1A). By day 7, when thrombus resolution is underway, treatment with each drug resulted in increased levels of active (nuclear) HIF1α in the thrombus and surrounding vein (Fig. 1B), along with increased levels of the HIF1 target, VEGF (Fig. 1C). Using proteome profiling, we next identified drug-specific signatures of pro-resolution proteins that were induced in the thrombus and vein (Fig. 1D). Treatment with each drug reduced thrombus length (Fig. 1E), weight (Fig. 1F), and volume (Fig. 1G, H) by >50% at day 7, which was accompanied in the Vadudustat and Daprodustat groups by >2-fold increases in vein recanalization (Fig. 1G, I). None of the drugs altered hematocrit at day 7 (Fig. 1J). To extend our safety assessments of the most effective drug, mice were pre-treated with Daprodustat for 6 days prior to thrombus induction (Fig. 2A). At day 1 post-thrombus induction, Daprodustat did not alter thrombus formation (length, Fig. 2B; or weight, Fig. 2C), the proteome profile of 53 circulating inflammatory cytokines (Fig. 2D), or inflammation (Fig. 2E-G) or vascular permeability (Fig. 2H-J) in the liver, kidney, or quadriceps. Finally, using a clinically equivalent regimen of an FDA-approved VEGF inhibitor (Bevacizumab, Fig. 3A, B), we showed that Daprodustat-driven increases in thrombus resolution are dependent upon VEGF (Fig. 3C).
CONCLUSIONS. Three different FDA-approved HIF1 agonists safely and effectively promote venous thrombus resolution in mice. Repurposing of FDA-approved HIF1 agonists represents a viable therapeutic option for the treatment of DVT.
APPROACH AND RESULTS. DVT was induced by inferior vena cava stenosis in male and female C57BL/6J mice (3-4m old). Mice were treated with oral vehicle or a clinically equivalent regimen of Rabeprazole (a novel HIF1 agonist), Vadadustat or Daprodustat (known HIF1 agonists). To assess efficacy, treatments were started at day 1 post-thrombus induction, when the thrombus is fully formed (Fig. 1A). By day 7, when thrombus resolution is underway, treatment with each drug resulted in increased levels of active (nuclear) HIF1α in the thrombus and surrounding vein (Fig. 1B), along with increased levels of the HIF1 target, VEGF (Fig. 1C). Using proteome profiling, we next identified drug-specific signatures of pro-resolution proteins that were induced in the thrombus and vein (Fig. 1D). Treatment with each drug reduced thrombus length (Fig. 1E), weight (Fig. 1F), and volume (Fig. 1G, H) by >50% at day 7, which was accompanied in the Vadudustat and Daprodustat groups by >2-fold increases in vein recanalization (Fig. 1G, I). None of the drugs altered hematocrit at day 7 (Fig. 1J). To extend our safety assessments of the most effective drug, mice were pre-treated with Daprodustat for 6 days prior to thrombus induction (Fig. 2A). At day 1 post-thrombus induction, Daprodustat did not alter thrombus formation (length, Fig. 2B; or weight, Fig. 2C), the proteome profile of 53 circulating inflammatory cytokines (Fig. 2D), or inflammation (Fig. 2E-G) or vascular permeability (Fig. 2H-J) in the liver, kidney, or quadriceps. Finally, using a clinically equivalent regimen of an FDA-approved VEGF inhibitor (Bevacizumab, Fig. 3A, B), we showed that Daprodustat-driven increases in thrombus resolution are dependent upon VEGF (Fig. 3C).
CONCLUSIONS. Three different FDA-approved HIF1 agonists safely and effectively promote venous thrombus resolution in mice. Repurposing of FDA-approved HIF1 agonists represents a viable therapeutic option for the treatment of DVT.
More abstracts on this topic:
A Silent Storm: Incidental Discovery of IVC and Right Atrium Thrombus in a Patient with Uterine Stromal Sarcoma
Wasef Natale, Fatima Tehreem, Stys Adam
A Rare Case of Loeffler Endocarditis and Intracardiac Thrombus in the setting of FIP1L1-PDGFRA MutationShaik Aleesha, Pankayatselvan Varayini, Mazar Michael, Bokhoor Pooya