Final ID: TH806
Outreach Strategy Impacts Trial Enrollment, Cost, Efficiency, and Diversity: Results from the GoFresh Trials
Abstract Body: Introduction
Approximately one-third of randomized controlled trials struggle to meet their recruitment goals. New recruitment strategies have emerged with the advent of online platforms, but their efficiency and cost-effectiveness vary by population. There is a critical need for more evidence on the effectiveness of diverse recruitment modalities, particularly for enrolling Black participants in clinical trials.
Objective
To identify differences in participant diversity, recruitment efficiency, and cost-effectiveness by recruitment modality for enrolling eligible trial participants.
Methods
The GoFresh trials tested whether DASH-patterned groceries might lower blood pressure (BP) in Black adults with elevated BP, residing in Boston-area food deserts. The trials employed various recruitment modalities and collectively screened over 5,000 adults. Recruitment data was collected during the screening phases of both trials. Odds ratios for recruiting participants with certain characteristics by recruitment modality were assessed using logistic regression, with word-of-mouth as the reference. Modality performance for randomization rates per 100 prescreened was evaluated with two-sample proportion tests.
Results
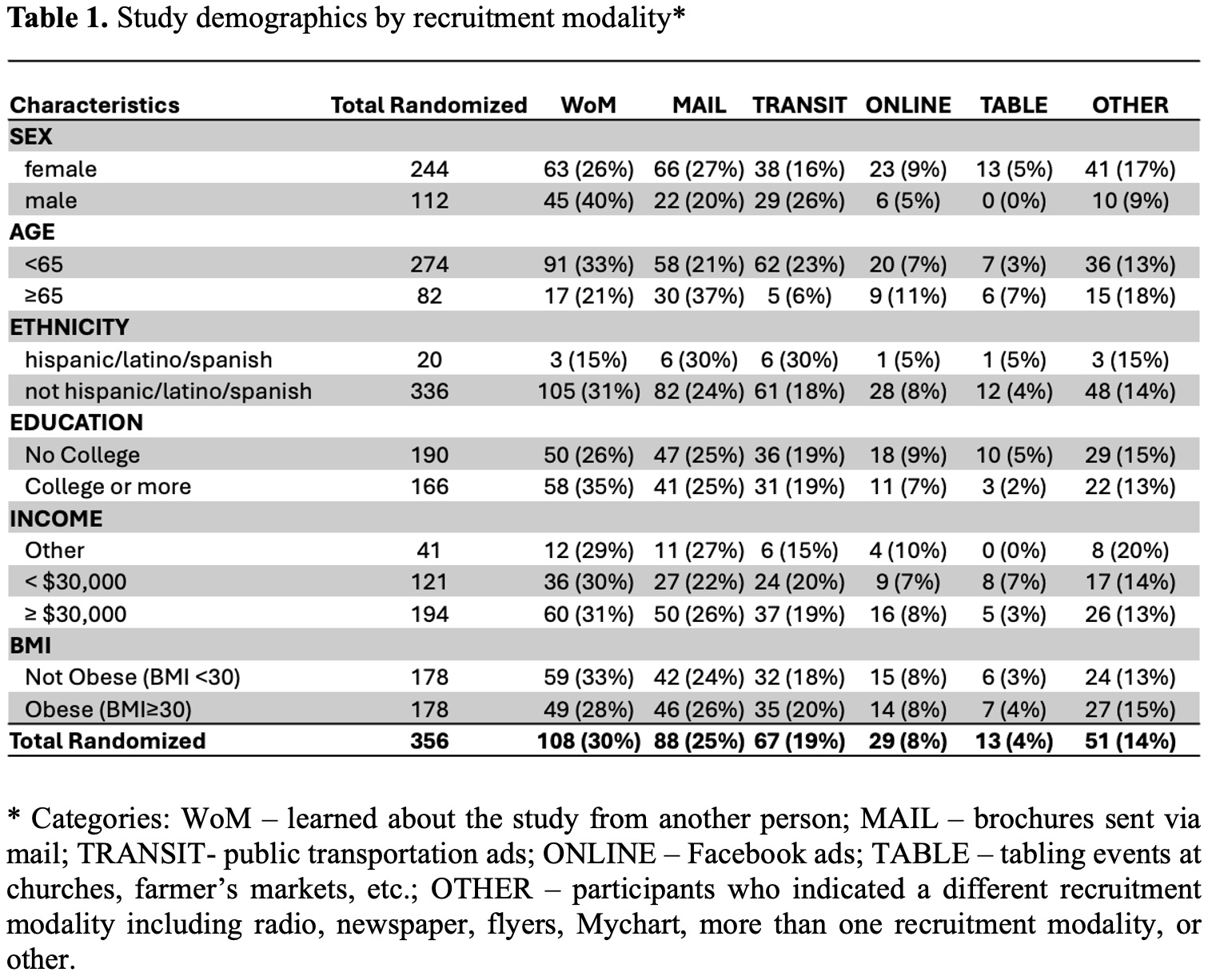
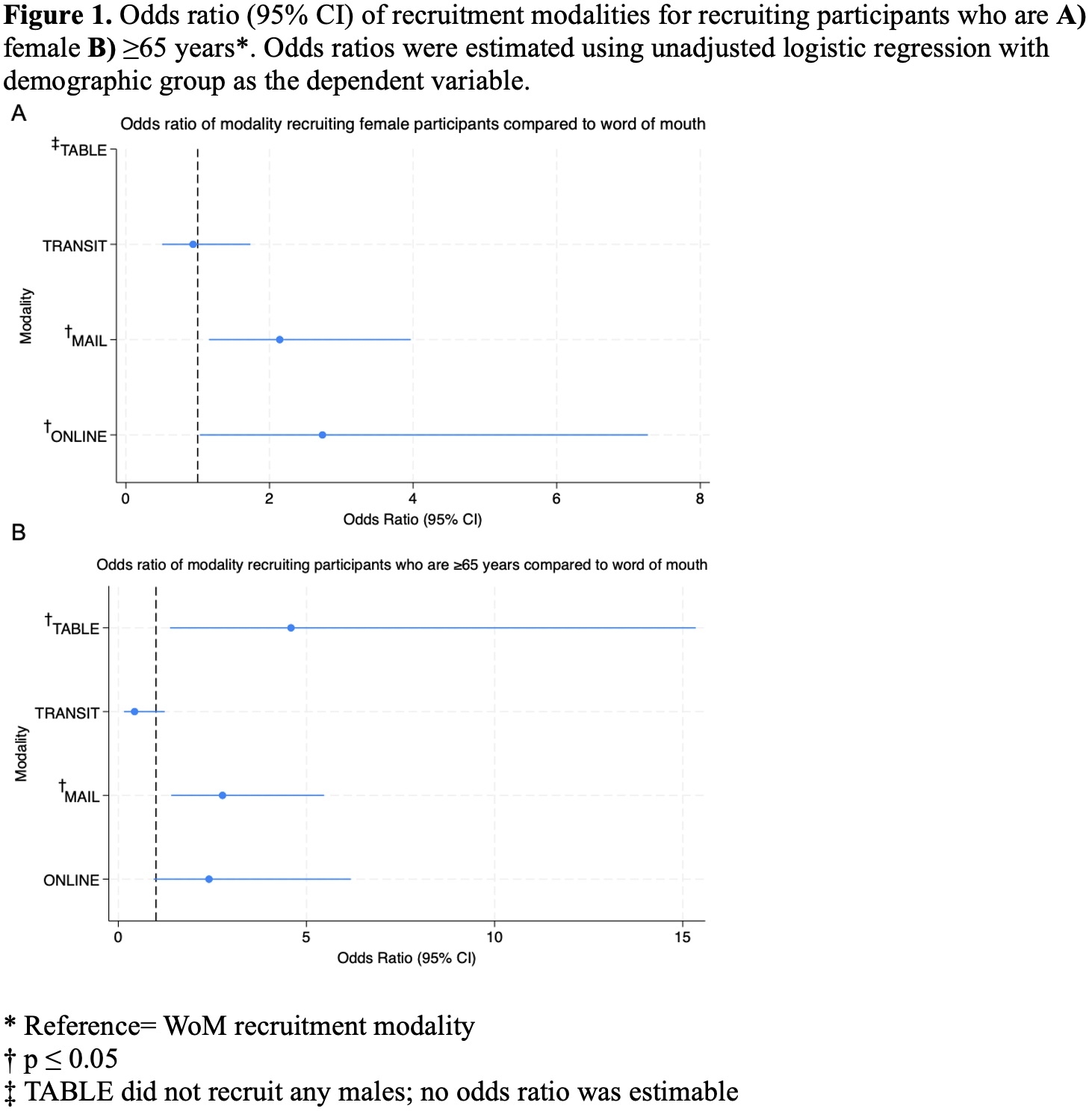
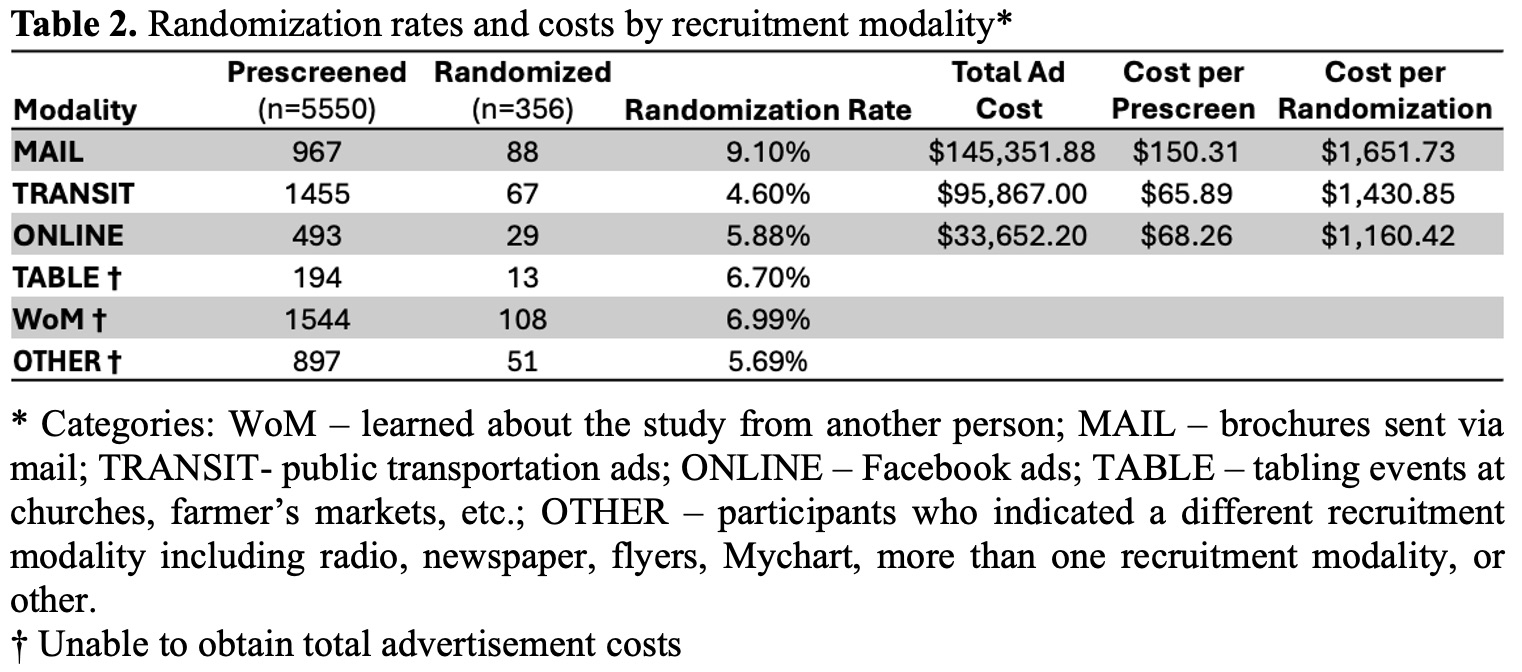
In this study of 356 participants, the top five recruitment modalities successfully enrolling participants were transit, mailing, tabling, online, and word-of-mouth (WoM) (Table 1). Compared to WoM, mail (OR 2.77, 95% CI: 1.40–5.47) and tabling (OR 4.59, 95% CI: 1.37–15.34) modalities were more likely to recruit participants ≥65 years (Figure 1). Compared to WoM, mail (OR 2.14, 95% CI: 1.16-3.97) and online (OR 2.74, 95% CI: 1.03-7.27) modalities were more likely to recruit female participants. Recruitment modalities did not differ significantly from WoM for recruiting college-educated, low-income, or obese participants. Mail had a significantly higher randomization rate than transit (difference in proportions: 4.5%, p<0.001) and online (difference in proportions 3.2%, p=0.03) modalities (Table 2). Cost per screening and per randomization varied by recruitment modality.
Conclusion
Mail, tabling, and online recruitment modalities were optimal for recruiting older female adults. Overall, recruitment strategies differed in efficiency and cost. Tailoring recruitment by modality can enhance enrollment success, resource use, and representation in clinical trials.
Approximately one-third of randomized controlled trials struggle to meet their recruitment goals. New recruitment strategies have emerged with the advent of online platforms, but their efficiency and cost-effectiveness vary by population. There is a critical need for more evidence on the effectiveness of diverse recruitment modalities, particularly for enrolling Black participants in clinical trials.
Objective
To identify differences in participant diversity, recruitment efficiency, and cost-effectiveness by recruitment modality for enrolling eligible trial participants.
Methods
The GoFresh trials tested whether DASH-patterned groceries might lower blood pressure (BP) in Black adults with elevated BP, residing in Boston-area food deserts. The trials employed various recruitment modalities and collectively screened over 5,000 adults. Recruitment data was collected during the screening phases of both trials. Odds ratios for recruiting participants with certain characteristics by recruitment modality were assessed using logistic regression, with word-of-mouth as the reference. Modality performance for randomization rates per 100 prescreened was evaluated with two-sample proportion tests.
Results
In this study of 356 participants, the top five recruitment modalities successfully enrolling participants were transit, mailing, tabling, online, and word-of-mouth (WoM) (Table 1). Compared to WoM, mail (OR 2.77, 95% CI: 1.40–5.47) and tabling (OR 4.59, 95% CI: 1.37–15.34) modalities were more likely to recruit participants ≥65 years (Figure 1). Compared to WoM, mail (OR 2.14, 95% CI: 1.16-3.97) and online (OR 2.74, 95% CI: 1.03-7.27) modalities were more likely to recruit female participants. Recruitment modalities did not differ significantly from WoM for recruiting college-educated, low-income, or obese participants. Mail had a significantly higher randomization rate than transit (difference in proportions: 4.5%, p<0.001) and online (difference in proportions 3.2%, p=0.03) modalities (Table 2). Cost per screening and per randomization varied by recruitment modality.
Conclusion
Mail, tabling, and online recruitment modalities were optimal for recruiting older female adults. Overall, recruitment strategies differed in efficiency and cost. Tailoring recruitment by modality can enhance enrollment success, resource use, and representation in clinical trials.
More abstracts on this topic:
A Phase I, Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of Single Ascending Doses of N-acetylgalactosamine Small Interfering RNA Conjugate, BPR-30221616, in Healthy Participants, for Potential Treatment of Transthyretin Amyloidosis
Han Xiaohong, Chen Rui, Cheng Xiwen, Zhang Langxi, Huang Haoxi, Fan Shengjun
A Phase 2 Study Evaluating the Effects of Mivelsiran, an Investigational RNA Interference Therapeutic, on Hemorrhagic and Nonhemorrhagic Manifestations of Cerebral Amyloid AngiopathyGreenberg Steven, Parikh Neal, Lee Jin-moo, Van Etten Ellis, Van Osch Matthias, Klijn Catharina, Sostelly Alexandre, Goteti Sasikiran, Sepehrband Farshid, Avbersek Andreja, Deering Robert