Final ID: TH886
Associations between Life’s Essential 8 and Plasma Biomarkers of Alzheimer’s Disease Pathology, Neurodegeneration, and Neuroinflammation across Mid- to Late-Life: The Atherosclerosis Risk in Communities Neurocognitive Study (ARIC-NCS)
Abstract Body: Introduction:
Emerging blood-based biomarkers provide a non-invasive and accessible means of quantifying dementia-related pathology and may help elucidate the biological mechanisms linking known cardiovascular risk factors and overall cardiovascular health to dementia risk. We tested the hypothesis that better midlife cardiovascular health, as measured by the American Heart Association's Life’s Essential 8 (LE8), is associated with lower levels of plasma biomarkers of neurodegeneration and neuroinflammation.
Methods:
Plasma biomarkers of amyloid beta (Aβ) 42 and 40, phosphorylated Tau-181 (p-Tau181), neurofilament light chain (NfL), and glial fibrillary acidic protein (GFAP) were measured using Quanterix Simoa immunoassays at Visits 3 (1993-1995), 5 (2011-2013), and 6/7 (2016-2019) in a sample of 1515 ARIC participants (Visit 3 mean age: 58.5 years, 39% male, 25.5% Black). Biomarker ratios (Aβ42:Aβ40, p-Tau181:Aβ42) and base-2 log transformed values of p-Tau181, NfL, and GFAP were derived and standardized to Visit 3 to facilitate comparisons between biomarkers. Visit 3 measures of diet, physical activity, smoking, sleep health, body mass index (BMI), blood lipids, blood glucose, and blood pressure were used to calculate a summary LE8 score (0-100) that was classified as low (0-49), moderate (50-79), or high (80-100), with higher values indicating better cardiovascular health. Covariate-adjusted linear mixed-effects models estimated associations of LE8 categories with 10-year rates of change in standardized biomarkers from 1993-2019 (median follow-up: 18.6 years). Attrition and selection weighting was applied to reduce potential bias.
Results:
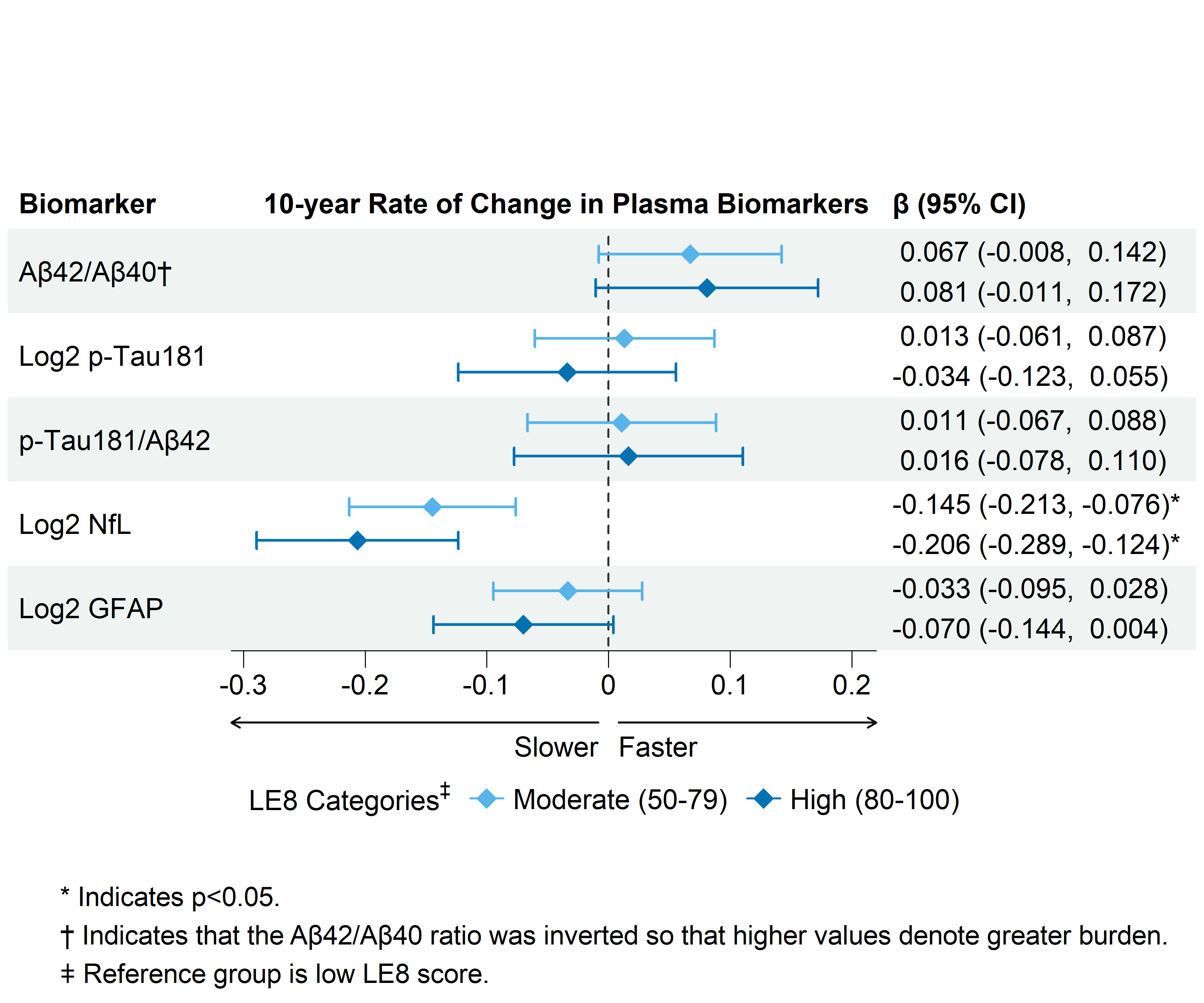
A slower rate of increase in NfL was observed among participants with moderate (Figure 1; β = -0.145, 95% CI: -0.213, -0.076) and high (β = -0.206, 95% CI: -0.289, -0.124) LE8 scores compared to those with low LE8 scores. Differences in the rates of change of other plasma biomarkers were not statistically significant.
Conclusions:
Better midlife LE8 scores were associated with slower increases in a biomarker of neurodegeneration. Midlife LE8 scores were not associated with biomarkers reflective of Alzheimer’s disease pathology. These findings suggest that the connection between LE8 measures in midlife and dementia decades later may be expressed in changes to biomarker levels, opening the door for tracking brain health changes through biomarkers of neurodegeneration.
Emerging blood-based biomarkers provide a non-invasive and accessible means of quantifying dementia-related pathology and may help elucidate the biological mechanisms linking known cardiovascular risk factors and overall cardiovascular health to dementia risk. We tested the hypothesis that better midlife cardiovascular health, as measured by the American Heart Association's Life’s Essential 8 (LE8), is associated with lower levels of plasma biomarkers of neurodegeneration and neuroinflammation.
Methods:
Plasma biomarkers of amyloid beta (Aβ) 42 and 40, phosphorylated Tau-181 (p-Tau181), neurofilament light chain (NfL), and glial fibrillary acidic protein (GFAP) were measured using Quanterix Simoa immunoassays at Visits 3 (1993-1995), 5 (2011-2013), and 6/7 (2016-2019) in a sample of 1515 ARIC participants (Visit 3 mean age: 58.5 years, 39% male, 25.5% Black). Biomarker ratios (Aβ42:Aβ40, p-Tau181:Aβ42) and base-2 log transformed values of p-Tau181, NfL, and GFAP were derived and standardized to Visit 3 to facilitate comparisons between biomarkers. Visit 3 measures of diet, physical activity, smoking, sleep health, body mass index (BMI), blood lipids, blood glucose, and blood pressure were used to calculate a summary LE8 score (0-100) that was classified as low (0-49), moderate (50-79), or high (80-100), with higher values indicating better cardiovascular health. Covariate-adjusted linear mixed-effects models estimated associations of LE8 categories with 10-year rates of change in standardized biomarkers from 1993-2019 (median follow-up: 18.6 years). Attrition and selection weighting was applied to reduce potential bias.
Results:
A slower rate of increase in NfL was observed among participants with moderate (Figure 1; β = -0.145, 95% CI: -0.213, -0.076) and high (β = -0.206, 95% CI: -0.289, -0.124) LE8 scores compared to those with low LE8 scores. Differences in the rates of change of other plasma biomarkers were not statistically significant.
Conclusions:
Better midlife LE8 scores were associated with slower increases in a biomarker of neurodegeneration. Midlife LE8 scores were not associated with biomarkers reflective of Alzheimer’s disease pathology. These findings suggest that the connection between LE8 measures in midlife and dementia decades later may be expressed in changes to biomarker levels, opening the door for tracking brain health changes through biomarkers of neurodegeneration.
More abstracts on this topic:
2-Methoxyestradiol By Inhibiting Central Action of 12S-Hydroxyeicosatetraenoic Acid Protects Ovariectomized Mice From Hypertension
Dutta Shubha, Singh Purnima, Song Chi Young, Shin Ji Soo, Malik Kafait
Association Between Health Care Access and Cardiovascular Health Risk Among Reproductive-Aged Women in the SAFE HEART StudyDankwa Kwabena, Vaidya Dhananjay, Ouyang Pamela, Hladek Melissa, Sharma Garima, Commodore-mensah Yvonne, Metlock Faith, Odei-kumi Kwabena, Ateh Stanislas Ketum, Rayani Asma, Baez Mateo Ana, Hernandez Lilian, Etelier Feodora, Ezuma Chioma