Final ID: TH827
Coffee Intake, Gut Microbiome, and Blood Metabolomic Pathways in Relation to Diabetes and Cardiometabolic Health: Findings from the Hispanic Community Health Study/Study of Latinos (HCHS/SOL)
Abstract Body: Background:
Coffee intake has been linked to improved cardiometabolic (CM) health, yet underlying microbial and metabolic pathways remain unclear.
Methods:
At HCHS/SOL Visit 1 (2008-11), usual coffee intake was estimated from two 24-hour dietary recalls and food propensity questionnaires among 13634 adults. Serum metabolomics (653 metabolites) were profiled by LC-MS at Visit 1 (n=6180) and Visit 2 (2014-17; n=718). Gut microbiome was characterized by shotgun metagenomics at Visit 2 (n=2758). CM traits, including adiposity, glycemic, lipid, blood pressure, kidney, and liver markers, were measured at both visits. Among 10730 participants free of diabetes, CVD, and cancer at baseline, 2493 diabetes cases occurred during a median follow-up of 10.2 years.
Results:
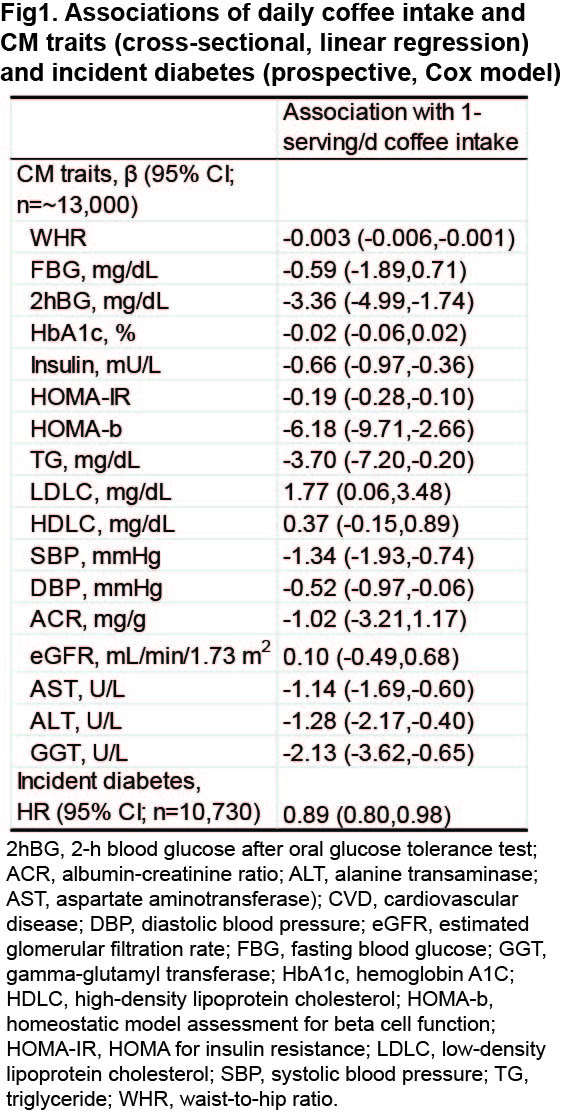
Higher coffee intake was correlated with lower levels of waist-to-hip ratio, liver enzymes (AST, ALT, GGT), glucose/insulin indices (2-h OGTT glucose, fasting insulin, HOMA-IR, HOMA-b), TG, and blood pressure, but higher LDLC levels (all P<.05). Each additional serving/d of coffee intake was related to an HR of 0.89 (95% CI 0.80-0.98; Fig1) for diabetes. Coffee intake was positively related to gut microbial α-diversity and four species (e.g., Clostridium phoceensis), while inversely related to 27 species, including pro-inflammatory Ruminococcus gnavus, Clostridium glycyrrhizinilyticum, and Blautia hansenii, and opportunistic Klebsiella oxytoca (q<.05), which were linked to unfavorable CM traits (Fig2).
We identified 165 coffee-related and 154 microbiota-related metabolites, with 55 overlapping. These 264 metabolites were clustered into 18 modules using WGCNA, one of which was mainly composed of caffeine metabolites and positively correlated with coffee intake and Clostridium phoceensis. A sphingomyelin module was positively related to coffee intake and lower diabetes risk (HR=0.88 [0.82-0.95] per SD increase in the module; Fig3). Modules of branched-chain amino acid (BCAA) and microbial bile acid metabolites were inversely related to coffee intake and higher diabetes risk, with HRs of 1.34 (1.24-1.45) and 1.07 (1.00-1.14), respectively. Glycerophospholipid module was positively related to Ruminococcus gnavus (inversely related to coffee intake) and an HR of 1.17 (1.09-1.26) for diabetes.
Conclusions:
Coffee intake was associated with gut microbial and metabolomics signatures converging on lipid, BCAA, and bile acid metabolism, and these pathways may illuminate the biological basis of coffee’s CM benefits.
Coffee intake has been linked to improved cardiometabolic (CM) health, yet underlying microbial and metabolic pathways remain unclear.
Methods:
At HCHS/SOL Visit 1 (2008-11), usual coffee intake was estimated from two 24-hour dietary recalls and food propensity questionnaires among 13634 adults. Serum metabolomics (653 metabolites) were profiled by LC-MS at Visit 1 (n=6180) and Visit 2 (2014-17; n=718). Gut microbiome was characterized by shotgun metagenomics at Visit 2 (n=2758). CM traits, including adiposity, glycemic, lipid, blood pressure, kidney, and liver markers, were measured at both visits. Among 10730 participants free of diabetes, CVD, and cancer at baseline, 2493 diabetes cases occurred during a median follow-up of 10.2 years.
Results:
Higher coffee intake was correlated with lower levels of waist-to-hip ratio, liver enzymes (AST, ALT, GGT), glucose/insulin indices (2-h OGTT glucose, fasting insulin, HOMA-IR, HOMA-b), TG, and blood pressure, but higher LDLC levels (all P<.05). Each additional serving/d of coffee intake was related to an HR of 0.89 (95% CI 0.80-0.98; Fig1) for diabetes. Coffee intake was positively related to gut microbial α-diversity and four species (e.g., Clostridium phoceensis), while inversely related to 27 species, including pro-inflammatory Ruminococcus gnavus, Clostridium glycyrrhizinilyticum, and Blautia hansenii, and opportunistic Klebsiella oxytoca (q<.05), which were linked to unfavorable CM traits (Fig2).
We identified 165 coffee-related and 154 microbiota-related metabolites, with 55 overlapping. These 264 metabolites were clustered into 18 modules using WGCNA, one of which was mainly composed of caffeine metabolites and positively correlated with coffee intake and Clostridium phoceensis. A sphingomyelin module was positively related to coffee intake and lower diabetes risk (HR=0.88 [0.82-0.95] per SD increase in the module; Fig3). Modules of branched-chain amino acid (BCAA) and microbial bile acid metabolites were inversely related to coffee intake and higher diabetes risk, with HRs of 1.34 (1.24-1.45) and 1.07 (1.00-1.14), respectively. Glycerophospholipid module was positively related to Ruminococcus gnavus (inversely related to coffee intake) and an HR of 1.17 (1.09-1.26) for diabetes.
Conclusions:
Coffee intake was associated with gut microbial and metabolomics signatures converging on lipid, BCAA, and bile acid metabolism, and these pathways may illuminate the biological basis of coffee’s CM benefits.
More abstracts on this topic:
A KLF2-BMPER-Smad1/5 checkpoint regulates high fluid shear stress-mediated artery remodeling
Deng Hanqiang, Zhang Jiasheng, Schwartz Martin
Cardiorenal Interaction Assessment via ECG Features: A Study using Dynamic Time Warping and Extracted Feature ClusteringZhao Sally, Adhin Bhavna, Zhan Ye, Fisch Sudeshna