Final ID: MPTH76
Genetic Propensity Modifies the Association Between Saturated Fat Intake and Low-Density Lipoprotein Cholesterol
Abstract Body: Introduction: The concentration of low-density lipoprotein cholesterol (LDL-C) in blood varies widely among individuals, and this variation is influenced by both genetics and diet. Clinical trials have implicated saturated fat (SFA) as a key macronutrient that raises LDL-C, yet individual responses to changes in SFA intake vary substantially. We investigated whether a polygenic score for LDL-C interacts with SFA intake to influence LDL-C.
Methods: This study included 20,940 genotyped participants of the Women’s Health Initiative with baseline LDL-C and food-frequency questionnaire (FFQ) data. A multi-ancestry polygenic score for LDL-C (PGS-LDL) was used to summarize genetic effects on LDL-C. The PGS-LDL was normalized by genetically inferred ancestry [European (EUR) = 10,488; African (AFR) = 7,046; American (AMR) = 2,996; East Asian = 410]. The effect of the PGS-LDL was modeled per 1 SD increase. Daily macronutrient intake was estimated from the FFQ. The effect of SFA was modeled as a 200 kcal/day increase of SFA at the expense of carbohydrates, using an isocaloric macronutrient substitution model. Multivariable linear regression was used to test the independent and interaction effects of the PGS-LDL and SFA on LDL-C. Models were adjusted for genetic PCs, genotyping array, and relevant clinical and lifestyle covariates. Both pooled and ancestry-stratified analyses were performed.
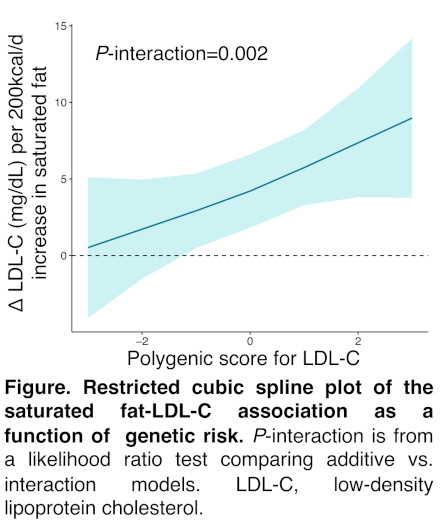
Results: The mean age was 64 ± 7 years. The mean LDL-C, adjusted for lipid-lowering therapy, was 152 ± 42 mg/dL. In a pooled analysis, the PGS-LDL and SFA were independently associated with LDL-C (PGS-LDL: 14.9 mg/dL, P < 2e-16; SFA: 4.3 mg/dL, P = 2e-4), and a significant PGS-LDL x SFA interaction was observed (P-interaction = 0.002). The magnitude of association between SFA and LDL-C became more pronounced with higher polygenic risk (Figure). This finding was consistent in stratified analyses among the three largest ancestry groups (AFR, AMR, EUR). In sensitivity analyses, we examined individuals without ASCVD (n = 19,483), without diabetes (n = 18,999), or not on lipid-lowering therapy (n = 19,049). We also repeated our analysis with an alternative EUR-derived PGS-LDL. All sensitivity analyses showed similar results (all P-interaction < 0.05).
Conclusions: Individuals with genetic propensity towards elevated LDL-C may be more sensitive to SFA intake. Further studies are needed to test these findings in clinical trials and evaluate their effects on cardiovascular disease risk.
Methods: This study included 20,940 genotyped participants of the Women’s Health Initiative with baseline LDL-C and food-frequency questionnaire (FFQ) data. A multi-ancestry polygenic score for LDL-C (PGS-LDL) was used to summarize genetic effects on LDL-C. The PGS-LDL was normalized by genetically inferred ancestry [European (EUR) = 10,488; African (AFR) = 7,046; American (AMR) = 2,996; East Asian = 410]. The effect of the PGS-LDL was modeled per 1 SD increase. Daily macronutrient intake was estimated from the FFQ. The effect of SFA was modeled as a 200 kcal/day increase of SFA at the expense of carbohydrates, using an isocaloric macronutrient substitution model. Multivariable linear regression was used to test the independent and interaction effects of the PGS-LDL and SFA on LDL-C. Models were adjusted for genetic PCs, genotyping array, and relevant clinical and lifestyle covariates. Both pooled and ancestry-stratified analyses were performed.
Results: The mean age was 64 ± 7 years. The mean LDL-C, adjusted for lipid-lowering therapy, was 152 ± 42 mg/dL. In a pooled analysis, the PGS-LDL and SFA were independently associated with LDL-C (PGS-LDL: 14.9 mg/dL, P < 2e-16; SFA: 4.3 mg/dL, P = 2e-4), and a significant PGS-LDL x SFA interaction was observed (P-interaction = 0.002). The magnitude of association between SFA and LDL-C became more pronounced with higher polygenic risk (Figure). This finding was consistent in stratified analyses among the three largest ancestry groups (AFR, AMR, EUR). In sensitivity analyses, we examined individuals without ASCVD (n = 19,483), without diabetes (n = 18,999), or not on lipid-lowering therapy (n = 19,049). We also repeated our analysis with an alternative EUR-derived PGS-LDL. All sensitivity analyses showed similar results (all P-interaction < 0.05).
Conclusions: Individuals with genetic propensity towards elevated LDL-C may be more sensitive to SFA intake. Further studies are needed to test these findings in clinical trials and evaluate their effects on cardiovascular disease risk.
More abstracts on this topic:
Diet quality, pathway-specific polygenic risk scores, and risk of type 2 diabetes among US men and women
Mei Zhendong, Liang Liming, Hu Frank, Li Jun, Wang Xingyan, Yun Huan, Sevilla-gonzalez Magdalena, Hu Jie, Bhupathiraju Shilpa, Sun Qi, Stampfer Meir, Willett Walter
A hepatic steatosis-mediated metabolite reprograms macrophage lipid metabolism and aggravates atherosclerosisLong Ting, Feng Ruijia, Feng Weiqi, Peng Guiyan, Yang Wenchao, Li Zilun, Huang Kan, Chang Guangqi