Final ID: MP1512
Artificial Intelligence-Enabled Electrocardiography Demonstrates Strong Diagnostic Performance for Diastolic Dysfunction, Heart Failure with Preserved Ejection Fraction, and Left Atrial Enlargement: A Meta-Analysis
Abstract Body (Do not enter title and authors here): Introduction:
Artificial intelligence (AI)-enabled electrocardiography (ECG) is redefining cardiac diagnostics by uncovering structural and functional abnormalities that human clinicians may not detect. Notably, AI models have shown promise in characterizing left ventricular diastolic dysfunction (LVDD), heart failure with preserved ejection fraction (HFpEF), and left atrial enlargement (LAE). However, the diagnostic performance of these models varies across studies. This meta-analysis evaluates the pooled performance of ECG-based AI models in detecting LVDD, HFpEF, and LAE.
Research Question:
What is the pooled diagnostic accuracy of AI-enabled ECG models for detecting (1) LVDD, (2) HFpEF, and (3) LAE?
Methods:
We systematically searched MEDLINE and Embase through May 2025 for studies evaluating AI-based ECG models for the diagnosis of LVDD, HFpEF, or LAE. Studies were included if they reported or allowed derivation of 2×2 confusion matrix data. A bivariate random-effects meta-analysis was conducted to pool sensitivity, specify, and area under the summary receiver operating characteristic (SROC) curve. Heterogeneity was quantified using sample size-adjusted I2 estimates.
Results:
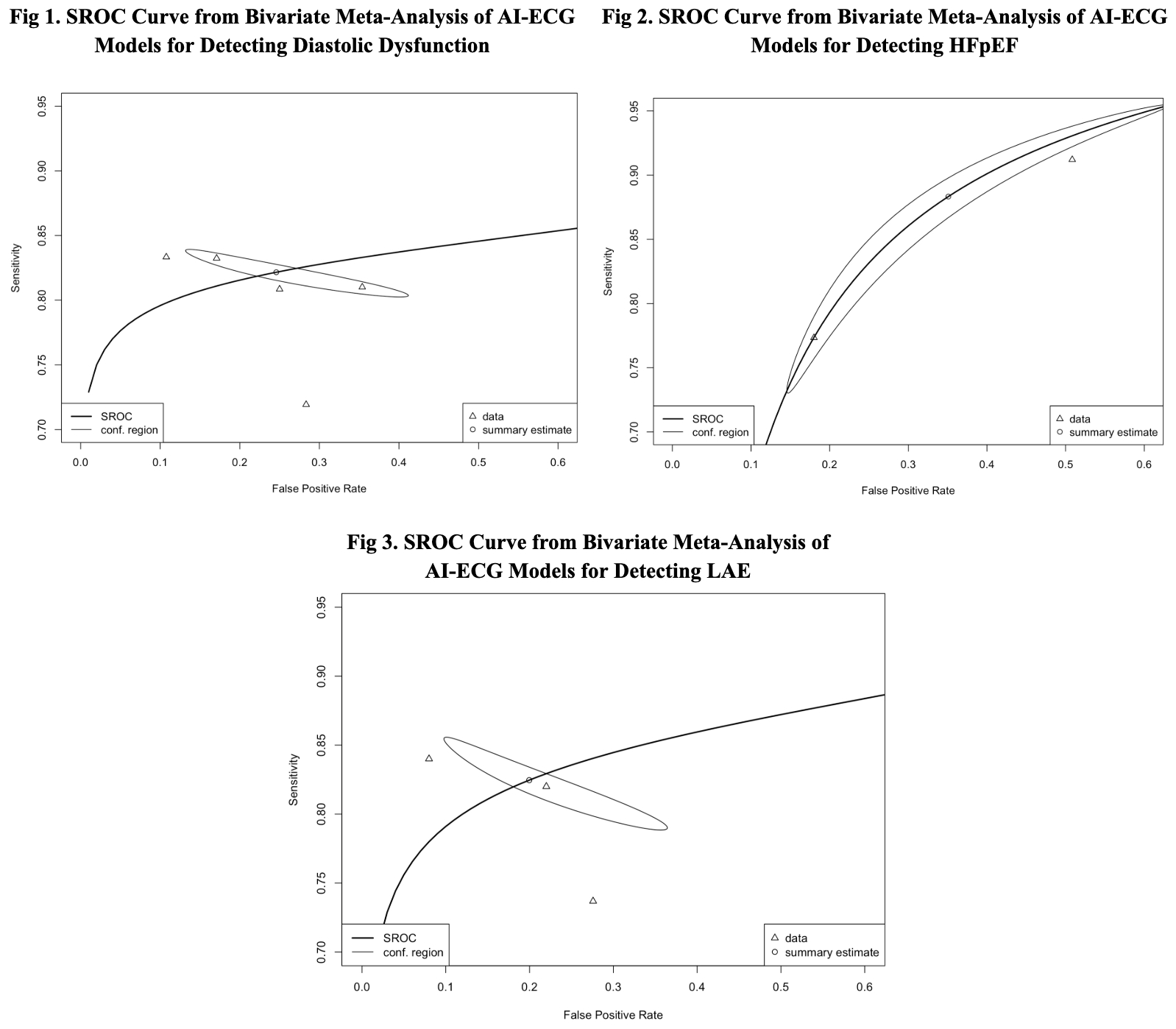
ECG-based AI models demonstrated strong diagnostic accuracy for LVDD, HFpEF, and LAE. Seven studies assessed models for detecting LVDD and yielded a pooled sensitivity of 0.82 (95% CI: 0.81–0.84) and a specificity of 0.75 (95% CI: 0.62–0.85), with an area under the SROC curve (AUC) of 0.83. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimates ranging from 1.5% to 3.1%. For HFpEF, three studies yielded a pooled sensitivity of 0.88 (95% CI: 0.77–0.95) and a specificity of 0.65 (95% CI: 0.42–0.82), with an AUC of 0.86. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimates ranging from 0.7% to 2.2%. Similarly, for left atrial enlargement (LAE), three studies yielded a pooled sensitivity of 0.83 (95% CI: 0.80–0.85) and a specificity of 0.80 (95% CI: 0.67–0.89), with an AUC of 0.86. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimated at 1.5%.
Conclusion:
AI-enabled ECG models demonstrate robust diagnostic performance for detecting LVDD, HFpEF, and LAE, with strong overall accuracy. These findings highlight the potential of AI as a scalable tool to enhance early detection of structural heart disease and support broader adoption of AI-driven screening in clinical practice.
Artificial intelligence (AI)-enabled electrocardiography (ECG) is redefining cardiac diagnostics by uncovering structural and functional abnormalities that human clinicians may not detect. Notably, AI models have shown promise in characterizing left ventricular diastolic dysfunction (LVDD), heart failure with preserved ejection fraction (HFpEF), and left atrial enlargement (LAE). However, the diagnostic performance of these models varies across studies. This meta-analysis evaluates the pooled performance of ECG-based AI models in detecting LVDD, HFpEF, and LAE.
Research Question:
What is the pooled diagnostic accuracy of AI-enabled ECG models for detecting (1) LVDD, (2) HFpEF, and (3) LAE?
Methods:
We systematically searched MEDLINE and Embase through May 2025 for studies evaluating AI-based ECG models for the diagnosis of LVDD, HFpEF, or LAE. Studies were included if they reported or allowed derivation of 2×2 confusion matrix data. A bivariate random-effects meta-analysis was conducted to pool sensitivity, specify, and area under the summary receiver operating characteristic (SROC) curve. Heterogeneity was quantified using sample size-adjusted I2 estimates.
Results:
ECG-based AI models demonstrated strong diagnostic accuracy for LVDD, HFpEF, and LAE. Seven studies assessed models for detecting LVDD and yielded a pooled sensitivity of 0.82 (95% CI: 0.81–0.84) and a specificity of 0.75 (95% CI: 0.62–0.85), with an area under the SROC curve (AUC) of 0.83. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimates ranging from 1.5% to 3.1%. For HFpEF, three studies yielded a pooled sensitivity of 0.88 (95% CI: 0.77–0.95) and a specificity of 0.65 (95% CI: 0.42–0.82), with an AUC of 0.86. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimates ranging from 0.7% to 2.2%. Similarly, for left atrial enlargement (LAE), three studies yielded a pooled sensitivity of 0.83 (95% CI: 0.80–0.85) and a specificity of 0.80 (95% CI: 0.67–0.89), with an AUC of 0.86. Between-study heterogeneity was minimal, with sample size–adjusted I2 estimated at 1.5%.
Conclusion:
AI-enabled ECG models demonstrate robust diagnostic performance for detecting LVDD, HFpEF, and LAE, with strong overall accuracy. These findings highlight the potential of AI as a scalable tool to enhance early detection of structural heart disease and support broader adoption of AI-driven screening in clinical practice.
More abstracts on this topic:
A Personal Risk Assessment Device in Patients with Chest Pain
Shvilkin Alexei, Zlatic Natasa, Atanasoski Vladimir, Grujovic Zdolsek Sanja, Popovic Maneski Lana, Miletic Marjan, Vukcevic Vladan
4-Hydroxy-2-Nonenal Alters Alternative Polyadenylation to Regulate mRNA Isoform Diversity in the Transition from Human Cardiac Fibroblasts to MyofibroblastsNatarajan Kartiga, Neupane Rahul, Yalamanchili Hari Krishna, Palaniyandi Suresh, Wagner Eric, Guha Ashrith, Amirthalingam Thandavarayan Rajarajan