Final ID: MP1723
Effect Of Inclisiran-based Treatment Strategy, In Combination With Individually Optimized Statin Therapy, On Quality Of Life And Muscle-related Pain vs. Standard of Care: Exploratory Outcomes From The VICTORION-Difference Study
Abstract Body (Do not enter title and authors here): Background: Inclisiran provides sustained and effective LDL-C reduction in individuals with hyperlipidemia with a favorable long-term safety profile; however, its impact on QoL remains unknown. The VICTORION-Difference study evaluated the early efficacy, safety, as well as QoL outcomes with inclisiran vs. placebo on top of individually optimized lipid-lowering therapy (ioLLT; standard of care [SOC]) in individuals with hypercholesterolemia who have not achieved guideline-recommended LDL-C goals.
Research question: Whether inclisiran-based treatment strategy vs. SOC reduces muscle-related adverse events (MRAEs) and days with pain experienced. Data on additional key secondary and exploratory endpoints for efficacy and safety, including LDL-C reduction, pain-related QoL scores, AEs and serious AEs will be presented.
Methods: This phase 4, double-blind, placebo-controlled trial included adults with hypercholesterolemia at high or very high CV risk on maximally tolerated dose (MTD) of statins. Participants were randomized 1:1 to receive subcutaneous injections of 300 mg inclisiran sodium or placebo on top of SOC at Days 1, 90, and 270. At randomization, participants received open-label rosuvastatin as background ioLLT; starting dose of 5 mg/day or 10 mg/day and sequentially titrated to the MTD. A logistic regression model analyzed the proportion of participants experiencing ≥1 MRAE from Day 1 to Day 360, while a negative binomial model assessed the annualized number of pain days. The study was powered for the key secondary endpoint related to MRAEs.
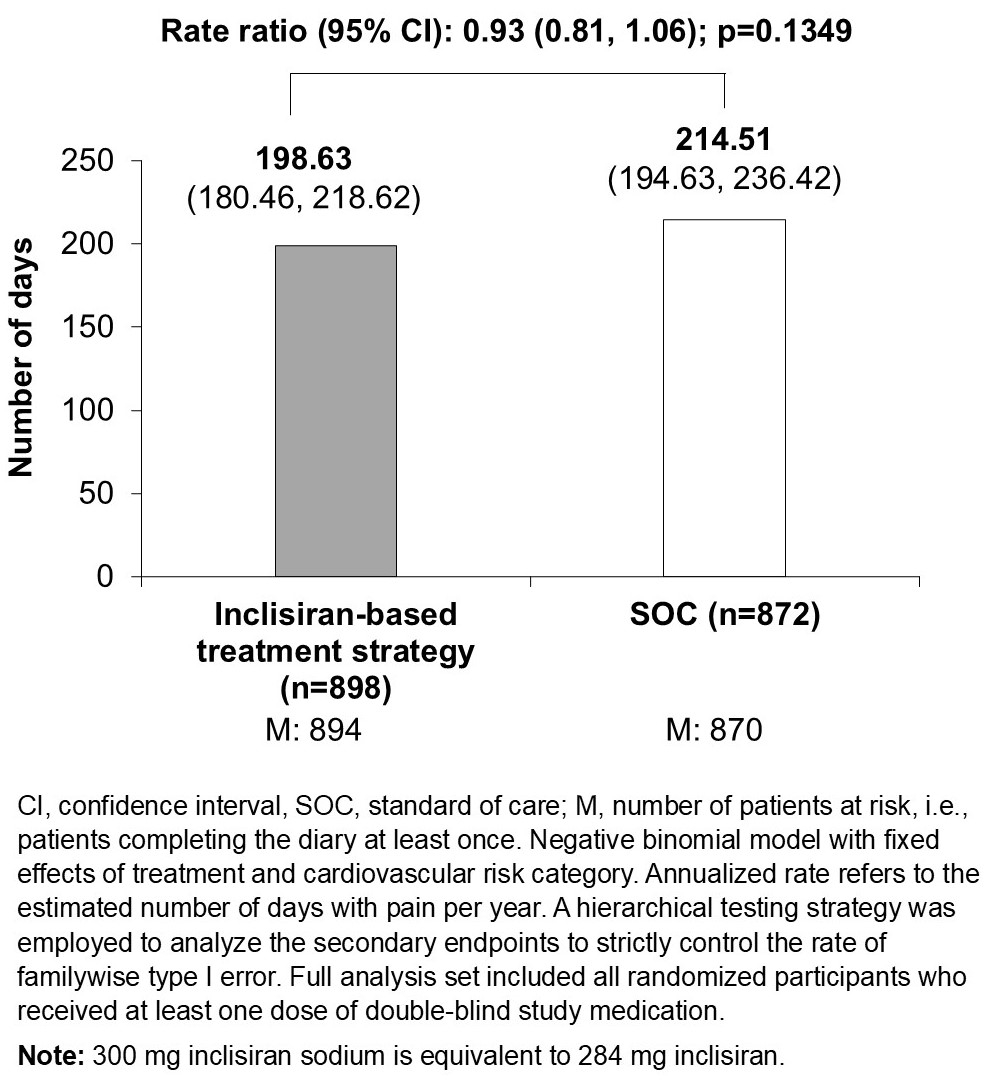
Results: A total of 1770 individuals (mean age, 63.7 years) were randomized to receive study medication (inclisiran, n=898; SOC, n=872), with 92.3% of participants classified as having very high CV risk. Fewer participants in the inclisiran vs. SOC arm experienced a MRAE (11.9% vs. 19.2%; odds ratio=0.57; 95% CI: 0.43, 0.74). Participants receiving inclisiran experienced numerically fewer days with pain vs. SOC (198.63 vs. 214.51; rate ratio: 0.93; 95% CI: 0.81, 1.06; Figure).
Conclusion: VICTORION-Difference is the largest LDL-C–lowering study from the inclisiran clinical development program so far, and the first to evaluate the effect of inclisiran vs. placebo on top of SOC using patient-centric QoL scores as secondary endpoints. Overall, inclisiran-based treatment strategy vs. SOC led to significantly fewer MRAEs and a numerical reduction in the number of days individuals with hypercholesterolemia experienced pain.
Research question: Whether inclisiran-based treatment strategy vs. SOC reduces muscle-related adverse events (MRAEs) and days with pain experienced. Data on additional key secondary and exploratory endpoints for efficacy and safety, including LDL-C reduction, pain-related QoL scores, AEs and serious AEs will be presented.
Methods: This phase 4, double-blind, placebo-controlled trial included adults with hypercholesterolemia at high or very high CV risk on maximally tolerated dose (MTD) of statins. Participants were randomized 1:1 to receive subcutaneous injections of 300 mg inclisiran sodium or placebo on top of SOC at Days 1, 90, and 270. At randomization, participants received open-label rosuvastatin as background ioLLT; starting dose of 5 mg/day or 10 mg/day and sequentially titrated to the MTD. A logistic regression model analyzed the proportion of participants experiencing ≥1 MRAE from Day 1 to Day 360, while a negative binomial model assessed the annualized number of pain days. The study was powered for the key secondary endpoint related to MRAEs.
Results: A total of 1770 individuals (mean age, 63.7 years) were randomized to receive study medication (inclisiran, n=898; SOC, n=872), with 92.3% of participants classified as having very high CV risk. Fewer participants in the inclisiran vs. SOC arm experienced a MRAE (11.9% vs. 19.2%; odds ratio=0.57; 95% CI: 0.43, 0.74). Participants receiving inclisiran experienced numerically fewer days with pain vs. SOC (198.63 vs. 214.51; rate ratio: 0.93; 95% CI: 0.81, 1.06; Figure).
Conclusion: VICTORION-Difference is the largest LDL-C–lowering study from the inclisiran clinical development program so far, and the first to evaluate the effect of inclisiran vs. placebo on top of SOC using patient-centric QoL scores as secondary endpoints. Overall, inclisiran-based treatment strategy vs. SOC led to significantly fewer MRAEs and a numerical reduction in the number of days individuals with hypercholesterolemia experienced pain.
More abstracts on this topic:
Advanced Care Planning in the CCU: What are the Numbers?
Sweeney Daniel, Uppal Priya, Jamrozik Thomas, Hutchison Paul
A multi-proteomic Risk Score Predicts Adverse Cardiovascular Outcomes in Patients with Angina and Non-obstructive Coronary Artery DiseaseHuang Jingwen, Lodhi Rafia, Lodhi Saleha, Eldaidamouni Ahmed, Hritani Wesam, Hasan Muhammet, Haroun Nisreen, Quyyumi Arshed, Mehta Puja, Leon Ana, Ko Yi-an, Yang Huiying, Medina-inojosa Jose, Ahmed Taha, Harris Kristen, Alkhoder Ayman, Al Kasem Mahmoud