Final ID: MDP12
Effect of mild hepatic impairment on the pharmacokinetics of pelacarsen
Abstract Body (Do not enter title and authors here): Introduction: Pelacarsen, a hepatocyte-directed, N-acetyl galactosamine (GalNAc3)–conjugated antisense oligonucleotide, reduces plasma lipoprotein(a) levels by inhibiting apolipoprotein(a) translation. Pelacarsen uptake is mediated by GalNAc3 binding to the hepatocyte-specific asialoglycoprotein receptor.
Hypothesis: It is unknown whether hepatic impairment (HI) impacts pelacarsen uptake and systemic exposure.
Aim: This single-dose, open-label, parallel-group, Phase 1 study (NCT05026996) assessed the pharmacokinetics (PK), safety, and tolerability of a single 80 mg subcutaneous dose of pelacarsen in participants with mild HI compared to healthy controls (normal hepatic function).
Methods: Eight adults with prior liver cirrhosis and mild HI (Child-Pugh Class A) were matched for sex, age, and body weight with nine healthy controls. PK parameters (Cmax, AUC0-72, AUClast, and AUCinf) were determined using non-compartmental methods. Log-transformed PK parameters were analyzed using a statistical model with group and matching covariates as fixed effects. Least-square geometric means for each group and geometric mean ratios between participants with mild HI and healthy controls were extracted. Safety was also assessed.
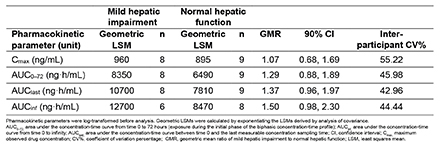
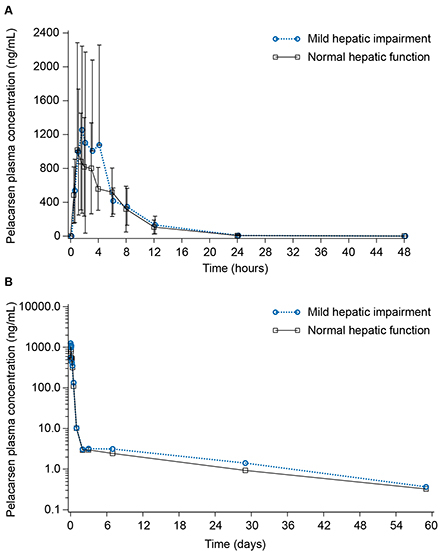
Results: Pelacarsen Cmax, AUClast, and AUCinf were, on average, 7%, 37%, and 50% higher, respectively, in participants with mild HI versus matched controls. All 90% confidence intervals around the HI versus healthy control geometric mean ratios included 1 (Table). The ranges of all PK parameters and estimated half-lives were similar between groups. In participants with mild HI, pelacarsen exposure approached the same level as controls after eight hours post-dose (Figure). No serious adverse events occurred.
Conclusion(s): In participants with mild HI, pelacarsen was well tolerated. Mild HI had no significant effect on pelacarsen Cmax. The non-statistically significant transient increase in AUC was within the exposure range tested in the first-in-human study.
Hypothesis: It is unknown whether hepatic impairment (HI) impacts pelacarsen uptake and systemic exposure.
Aim: This single-dose, open-label, parallel-group, Phase 1 study (NCT05026996) assessed the pharmacokinetics (PK), safety, and tolerability of a single 80 mg subcutaneous dose of pelacarsen in participants with mild HI compared to healthy controls (normal hepatic function).
Methods: Eight adults with prior liver cirrhosis and mild HI (Child-Pugh Class A) were matched for sex, age, and body weight with nine healthy controls. PK parameters (Cmax, AUC0-72, AUClast, and AUCinf) were determined using non-compartmental methods. Log-transformed PK parameters were analyzed using a statistical model with group and matching covariates as fixed effects. Least-square geometric means for each group and geometric mean ratios between participants with mild HI and healthy controls were extracted. Safety was also assessed.
Results: Pelacarsen Cmax, AUClast, and AUCinf were, on average, 7%, 37%, and 50% higher, respectively, in participants with mild HI versus matched controls. All 90% confidence intervals around the HI versus healthy control geometric mean ratios included 1 (Table). The ranges of all PK parameters and estimated half-lives were similar between groups. In participants with mild HI, pelacarsen exposure approached the same level as controls after eight hours post-dose (Figure). No serious adverse events occurred.
Conclusion(s): In participants with mild HI, pelacarsen was well tolerated. Mild HI had no significant effect on pelacarsen Cmax. The non-statistically significant transient increase in AUC was within the exposure range tested in the first-in-human study.
More abstracts on this topic:
Pharmacokinetics and pharmacodynamics of SC furosemide in patients with HF and obesity
Alexy Tamas, Kamineni Phani, Harata Mikako, Luepke Katie, Goodwin Matthew, Mohr John
Acute Administration of The Novel Cardiac Sarcomere Modulator EDG-7500, Improves Ventricular Filling While Preserving LVEF In Dogs with Pacing Induced Left-Ventricular Systolic DysfunctionEvanchik Marc, Emter Craig, Del Rio Carlos, Roof Steve, St Clair Sydney, Russell Alan, Henze Marcus, Semigran Marc