Final ID: MDP740
Checkpoint Kinase 1 Attenuates Myocardial Ischemia-Reperfusion Injury Through Maintaining SIRT1-Dependent Mitochondrial Homeostasis
Abstract Body (Do not enter title and authors here): Background: Mitochondrial dysfunction is linked to myocardial ischemia-reperfusion (I/R) injury. Checkpoint kinase 1 (CHK1) could facilitate cardiomyocyte proliferation and cardiac repair post myocardial infarction, however, its role on mitochondrial function in I/R injury remains unknown.
Research Questions: The purpose of this study is to explore the potential effects and mechanisms of CHK1 on mitochondrial homeostasis during myocardial I/R injury.
Methods: To investigate the role of CHK1 on mitochondrial function following I/R injury, cardiomyocyte-specific knockout/knock-in mouse models were generated. Mass spectrometry-proteomics analysis and protein co-immunoprecipitation assays were conducted to dissect the molecular mechanism of CHK1. The interaction between CHK1 and SIRT1 was explored using truncated plasmids.
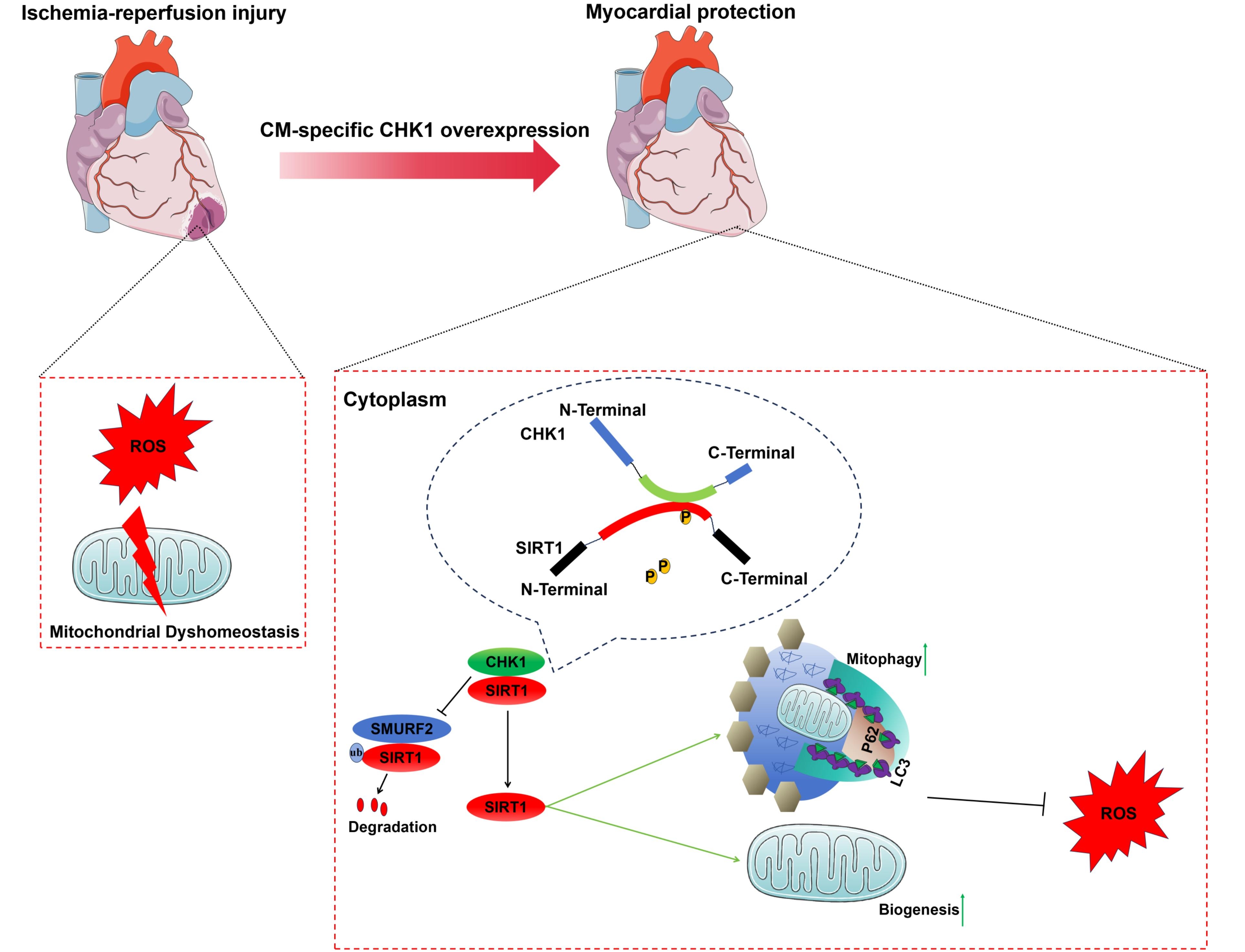
Results: CHK1 was downregulated in myocardium post I/R and neonatal mouse cardiomyocytes (NMCMs) post oxygen-glucose deprivation/re-oxygenation (OGD/R). In vivo, CHK1 overexpression protected against myocardial I/R injury, while heterogenous CHK1 knockout exacerbated cardiomyopathy. In vitro, CHK1 inhibited OGD/R-induced cardiomyocyte apoptosis and bolstered cardiomyocyte survival. Mechanistically, CHK1 attenuated oxidative stress and preserved mitochondrial metabolism in cardiomyocytes under I/R. Moreover, disrupted mitochondrial homeostasis in I/R myocardium was restored by CHK1 through the promotion of mitochondrial biogenesis and mitophagy. Through mass spectrometry analysis following co-immunoprecipitation, SIRT1 was identified as a direct target of CHK1. The 266-390 domain of CHK1 interacted with the 160-583 domain of SIRT1. Importantly, CHK1 phosphorylated SIRT1 at Thr530 residue, thereby inhibiting SMURF2-mediated degradation of SIRT1. CHK1 Δ390 amino acids (aa) mutant functioned similarly to full-length CHK1 in scavenging ROS and maintaining mitochondrial dynamics. Consistently, cardiac-specific SIRT1 knockdown partially attenuated the protective role of CHK1 in I/R injury.
Conclusions: Our findings revealed that CHK1 mitigates I/R injury and restores mitochondrial dynamics in cardiomyocytes through a SIRT1-dependent mechanism.
Research Questions: The purpose of this study is to explore the potential effects and mechanisms of CHK1 on mitochondrial homeostasis during myocardial I/R injury.
Methods: To investigate the role of CHK1 on mitochondrial function following I/R injury, cardiomyocyte-specific knockout/knock-in mouse models were generated. Mass spectrometry-proteomics analysis and protein co-immunoprecipitation assays were conducted to dissect the molecular mechanism of CHK1. The interaction between CHK1 and SIRT1 was explored using truncated plasmids.
Results: CHK1 was downregulated in myocardium post I/R and neonatal mouse cardiomyocytes (NMCMs) post oxygen-glucose deprivation/re-oxygenation (OGD/R). In vivo, CHK1 overexpression protected against myocardial I/R injury, while heterogenous CHK1 knockout exacerbated cardiomyopathy. In vitro, CHK1 inhibited OGD/R-induced cardiomyocyte apoptosis and bolstered cardiomyocyte survival. Mechanistically, CHK1 attenuated oxidative stress and preserved mitochondrial metabolism in cardiomyocytes under I/R. Moreover, disrupted mitochondrial homeostasis in I/R myocardium was restored by CHK1 through the promotion of mitochondrial biogenesis and mitophagy. Through mass spectrometry analysis following co-immunoprecipitation, SIRT1 was identified as a direct target of CHK1. The 266-390 domain of CHK1 interacted with the 160-583 domain of SIRT1. Importantly, CHK1 phosphorylated SIRT1 at Thr530 residue, thereby inhibiting SMURF2-mediated degradation of SIRT1. CHK1 Δ390 amino acids (aa) mutant functioned similarly to full-length CHK1 in scavenging ROS and maintaining mitochondrial dynamics. Consistently, cardiac-specific SIRT1 knockdown partially attenuated the protective role of CHK1 in I/R injury.
Conclusions: Our findings revealed that CHK1 mitigates I/R injury and restores mitochondrial dynamics in cardiomyocytes through a SIRT1-dependent mechanism.
More abstracts on this topic:
A Selective Agonist of the Beta 2 Retinoic Acid Receptor Reduces Necrosis, Lipid Peroxidation, and Oxidative Stress in Doxorubicin-induced Cardiotoxicity
Varzideh Fahimeh, Jankauskas Stanislovas, Kansakar Urna, Tang Xiao-han, Gudas Lorraine, Levi Roberto, Gambardella Jessica, Santulli Gaetano
Brown Adipose Tissue-Heart Crosstalk Attenuates Adverse Cardiac RemodelingShi Tingting, Chen Yang, Che Wenliang, Xiang Yaozu