Final ID: Su2086
Catheter Ablation for Ventricular Arrhythmias in Transthyretin Cardiac Amyloidosis: A Single Center Experience
Abstract Body (Do not enter title and authors here): Background: Transthyretin cardiac amyloidosis (ATTR-CA) carries an elevated risk of developing ventricular arrhythmias (VA) but there remains limited evidence supporting the role of catheter ablation in this population.
Purpose: To assess the safety and efficacy of catheter ablation for VA in ATTR-CA.
Methods: An electronic database was used to identify all patients suspected of having ATTR-CA who underwent catheter ablation for VA at our institution between January 2010 and December 2023. Patients who did not meet the diagnosis of ATTR-CA and had a negative workup who underwent catheter ablation for ventricular tachycardia (VT) or premature ventricular contractions (PVCs) were used to form a control group matched for age, sex and left ventricular ejection fraction at a ratio of 2:1.
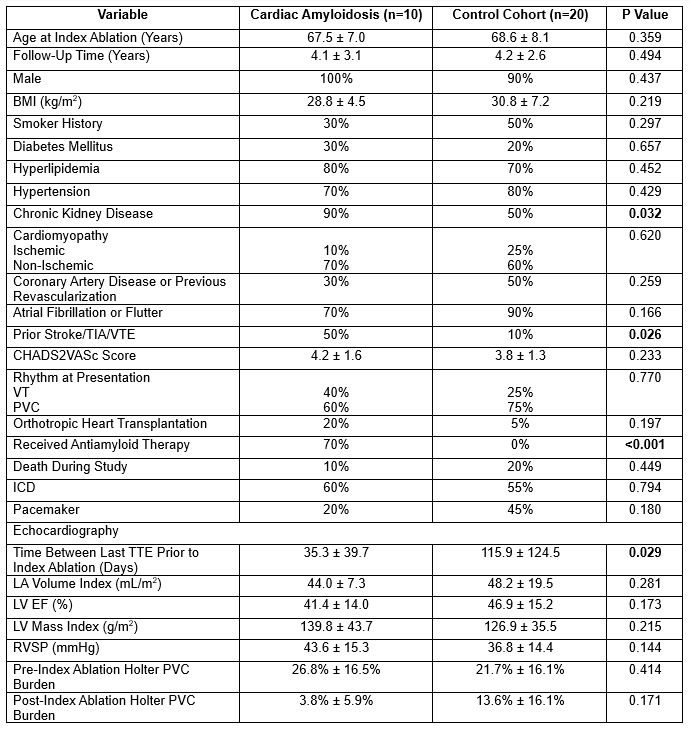
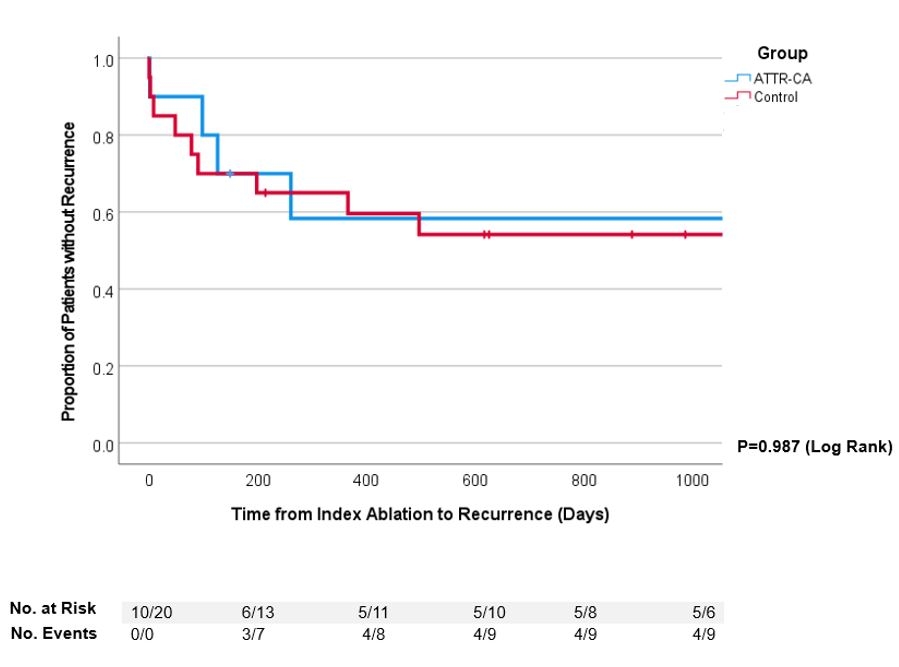
Results: 20 controls and 10 cases of ATTR-CA were identified. Examining the two groups at baseline (Table 1), the mean age at index ablation was 67.7±7.0 and 68.6±8.1 years (p=0.359), mean follow-up time was 4.1±3.1 and 4.2±2.6 years (p=0.494), and the proportion of males was 100% and 90% respectively. The ATTR-CA group had a significantly higher proportion of chronic kidney disease (90% vs. 50%, p=0.032) and prior stroke or thrombotic events compared to the control group (50% vs. 10%, p=0.026) with similar CHA2DS2VASc scores (4.2±1.6 vs. 3.8±1.3, p=0.233). Both groups had comparable left ventricular ejection fraction (41.4%±14% vs. 46.9%±15.2%, p=0.173), VT/PVCs at presentation (40%/60% vs. 25%/75%, p=0.77), use of antiarrhythmics (70% vs. 60%, p=0.592), and high proportion of atrial fibrillation/flutter (70% vs. 90%, p=0.168). Procedural (261.2±101.2 vs. 265.7±125.6 mins, p=0.449) and total energy delivery time (1442.9±1509.1 vs. 1744.6±956.2 secs, p=0.296) was not significantly different in the two groups. Both groups had a significant decrease in PVC burden postablation with a reduction from 26.8% to 3.8% (p=0.024) and from 27.1% to 13.6% (p=0.028) in the ATTR-CA and control group respectively. A downward trend was seen in antitachycardia pacing events for VT postablation in the ATTR-CA (14.7±9.8 vs. 5.3±6.7, p =0.184) and control (193±387.6 vs. 0.67±1.2. p=0.139) groups. No major complications occurred after ablation (Table 2). Mean time to recurrence post-index ablation was similar between both groups (1620.4±483.8 vs. 1498.8±291.4 days, p=0.987, Figure 1).
Conclusions: Catheter ablation appears to be safe and effective for VT and PVCs in patients with ATTR-CA.
Purpose: To assess the safety and efficacy of catheter ablation for VA in ATTR-CA.
Methods: An electronic database was used to identify all patients suspected of having ATTR-CA who underwent catheter ablation for VA at our institution between January 2010 and December 2023. Patients who did not meet the diagnosis of ATTR-CA and had a negative workup who underwent catheter ablation for ventricular tachycardia (VT) or premature ventricular contractions (PVCs) were used to form a control group matched for age, sex and left ventricular ejection fraction at a ratio of 2:1.
Results: 20 controls and 10 cases of ATTR-CA were identified. Examining the two groups at baseline (Table 1), the mean age at index ablation was 67.7±7.0 and 68.6±8.1 years (p=0.359), mean follow-up time was 4.1±3.1 and 4.2±2.6 years (p=0.494), and the proportion of males was 100% and 90% respectively. The ATTR-CA group had a significantly higher proportion of chronic kidney disease (90% vs. 50%, p=0.032) and prior stroke or thrombotic events compared to the control group (50% vs. 10%, p=0.026) with similar CHA2DS2VASc scores (4.2±1.6 vs. 3.8±1.3, p=0.233). Both groups had comparable left ventricular ejection fraction (41.4%±14% vs. 46.9%±15.2%, p=0.173), VT/PVCs at presentation (40%/60% vs. 25%/75%, p=0.77), use of antiarrhythmics (70% vs. 60%, p=0.592), and high proportion of atrial fibrillation/flutter (70% vs. 90%, p=0.168). Procedural (261.2±101.2 vs. 265.7±125.6 mins, p=0.449) and total energy delivery time (1442.9±1509.1 vs. 1744.6±956.2 secs, p=0.296) was not significantly different in the two groups. Both groups had a significant decrease in PVC burden postablation with a reduction from 26.8% to 3.8% (p=0.024) and from 27.1% to 13.6% (p=0.028) in the ATTR-CA and control group respectively. A downward trend was seen in antitachycardia pacing events for VT postablation in the ATTR-CA (14.7±9.8 vs. 5.3±6.7, p =0.184) and control (193±387.6 vs. 0.67±1.2. p=0.139) groups. No major complications occurred after ablation (Table 2). Mean time to recurrence post-index ablation was similar between both groups (1620.4±483.8 vs. 1498.8±291.4 days, p=0.987, Figure 1).
Conclusions: Catheter ablation appears to be safe and effective for VT and PVCs in patients with ATTR-CA.
More abstracts on this topic:
A-band titin-truncating variant promotes the development of arrhythmia-induced cardiomyopathy in a novel genetically-engineered porcine model
Lee Kwonjae, Del Rio Carlos, Mcnally Elizabeth, Pfenniger Anna, Bhatnagar Ashita, Glinton Kristofor, Burrell Amy, Ober Rebecca, Mcluckie Alicia, Bishop Brian, Rogers Christopher, Geist Gail
A Curious Complete Heart Block with CarfilzomibShah Mohammed, Rahman Naveed, Al-mohamad Talal, Batra Sejal, Vyas Apurva