Final ID: Wed155
De novo Protein N-Glycosylation in Platelets Regulates Platelet-T Cell Interactions
Abstract Body: Platelets are traditionally viewed as translationally and post-translationally limited due to their anucleate nature and lack of a canonical rough endoplasmic reticulum (ER) and stacked Golgi apparatus. However, accumulating evidence indicates that platelets are capable of de novo protein glycosylation. We intestigated this in our current study.
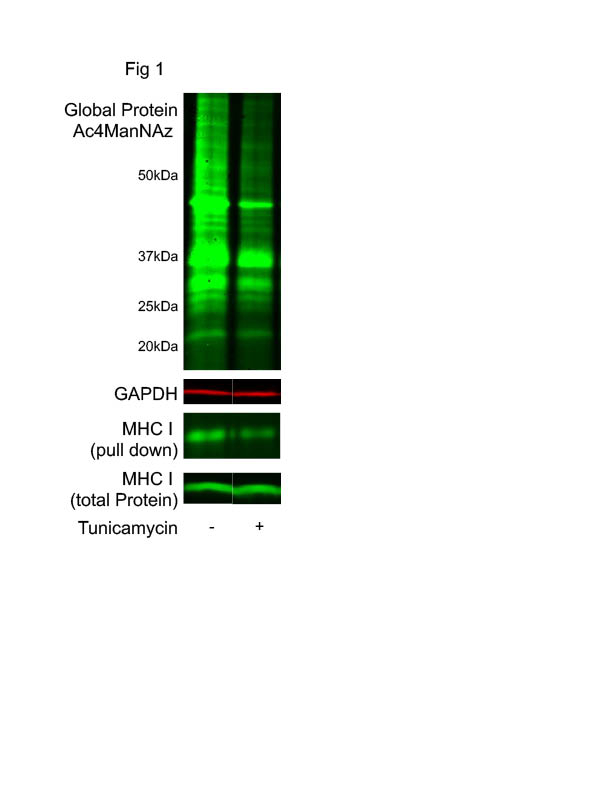
Platelets isolated from healthy donors or mice were incubated in vitro with azide-labeled monosaccharide precursors (Ac4ManNAz, 50μM) for 16 h at 37 °C. Incorporation of Ac4ManNAz into newly glycosylated proteins was detected by azide–alkyne reaction (Click-iT chemistry), revealing robust de novo glycosylation in platelets (Fig.1) Treatment with tunicamycin (5 µg/mL), a specific inhibitor of N-linked glycosylation, significantly reduced azide incorporation. Platelet viability following tunicamycin treatment was confirmed by negative annexin V staining.
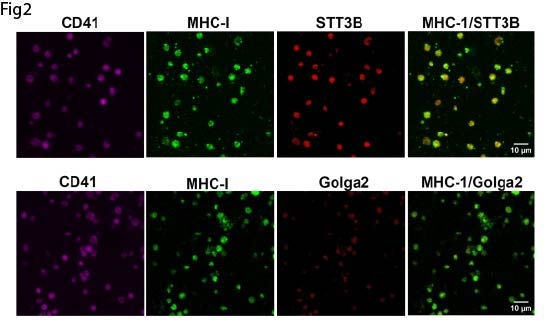
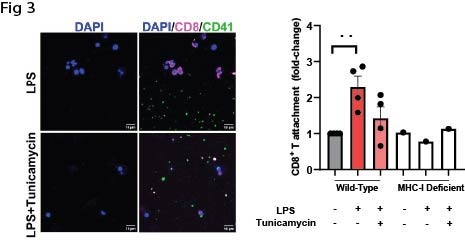
We next examined N-glycosylation of platelet major histocompatibility complex class I (MHC I), a receptor we previously identified as upregulated during thromboinflammation and critical for platelet–CD8+ T cell interactions. Azide-labeled glycoproteins were biotinylated, purified, and immunoblotted for MHC I. De novo glycosylated MHC I was detected and was inhibited by tunicamycin (Fig1). Consistent with this, human and mouse platelets expressed key components of the glycosylation machinery, including the oligosaccharyltransferase subunit STT3B and the Golgi matrix protein Golga2, which colocalized with MHC I (Fig2). N-glycosylation at Asn86 of the MHC I heavy chain is essential for proper assembly and trafficking. In platelets, tunicamycin treatment significantly reduced both total and surface MHC I expression, indicating a functional requirement for de novo N-glycosylation of MHC I. Analysis of platelet RNA-seq and published proteomic datasets revealed expression of the glycosyltransferases required for synthesis of the core Glc1Man9GlcNAc2 N-glycan. However, platelet MHC I exhibited a distinct molecular weight compared to leukocyte MHC I from the same donors, suggesting limited glycan maturation. Functionally, inhibition of N-glycosylation significantly impaired MHC I–dependent binding between antigen-pulsed platelets and cognate CD8+T cells (Fig 3).
Together, we found that platelets possess an active N-glycosylation pathway, generating distinct glycoforms that modulate platelet immune functions, including antigen presentation and T cell interactions.
Platelets isolated from healthy donors or mice were incubated in vitro with azide-labeled monosaccharide precursors (Ac4ManNAz, 50μM) for 16 h at 37 °C. Incorporation of Ac4ManNAz into newly glycosylated proteins was detected by azide–alkyne reaction (Click-iT chemistry), revealing robust de novo glycosylation in platelets (Fig.1) Treatment with tunicamycin (5 µg/mL), a specific inhibitor of N-linked glycosylation, significantly reduced azide incorporation. Platelet viability following tunicamycin treatment was confirmed by negative annexin V staining.
We next examined N-glycosylation of platelet major histocompatibility complex class I (MHC I), a receptor we previously identified as upregulated during thromboinflammation and critical for platelet–CD8+ T cell interactions. Azide-labeled glycoproteins were biotinylated, purified, and immunoblotted for MHC I. De novo glycosylated MHC I was detected and was inhibited by tunicamycin (Fig1). Consistent with this, human and mouse platelets expressed key components of the glycosylation machinery, including the oligosaccharyltransferase subunit STT3B and the Golgi matrix protein Golga2, which colocalized with MHC I (Fig2). N-glycosylation at Asn86 of the MHC I heavy chain is essential for proper assembly and trafficking. In platelets, tunicamycin treatment significantly reduced both total and surface MHC I expression, indicating a functional requirement for de novo N-glycosylation of MHC I. Analysis of platelet RNA-seq and published proteomic datasets revealed expression of the glycosyltransferases required for synthesis of the core Glc1Man9GlcNAc2 N-glycan. However, platelet MHC I exhibited a distinct molecular weight compared to leukocyte MHC I from the same donors, suggesting limited glycan maturation. Functionally, inhibition of N-glycosylation significantly impaired MHC I–dependent binding between antigen-pulsed platelets and cognate CD8+T cells (Fig 3).
Together, we found that platelets possess an active N-glycosylation pathway, generating distinct glycoforms that modulate platelet immune functions, including antigen presentation and T cell interactions.
More abstracts on this topic:
A meta-analysis of RNA sequencing data to characterize immune cell activation among patients with cardiac sarcoidosis
Banga Natasha, Huang Yuhsin, Chou Harrison, Vitale Maria Rosaria, Baker Matthew, Witteles Ronald, Zhu Han
Activator protein 1 (AP-1) Complex in Antigen Presenting Cells contributes to Salt-Sensitive Blood Pressure in HumansAhmad Taseer, Desta Selam, Kirabo Annet, Saleem Mohammad, Mutchler Ashley Pitzer, Ertuglu Lale, Albritton Claude, Haynes Alexandria, Sheng Quanhu, Khan Mohd, Demirci Mert