Final ID: Thu101
Mechanotransduction and Endpoint Mechanisms of Ultrasound-Enhanced Vascular Permeability
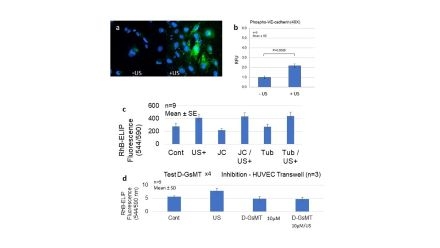
Abstract Body: Introduction. We have used ultrasound (US) to facilitate penetration of echogenic liposome (ELIP) formulations into arterial walls for diagnostic and therapeutic applications. Mechanisms responsible for this US-enhanced vascular permeability are not well understood. In a previous study, we found that the phenomenon is caused by radiation pressure, rather than cavitation effects, and that it is mediated by nitric oxide (NO) signaling pathways featuring Akt and extracellular-regulated kinase (ERK) 1/2 phosphorylation. We have now determined the nature of the initiation signal and endpoint mechanism mediating the effect. Hypotheses. A Piezo mechanotransducer and VE- cadherin are responsible for initiating and effecting US-enhanced vascular permeability, respectively. Methods. Human umbilical vein endothelial cells (HUVEC) grown on 4-chamber culture slides, with and without continuous-wave US treatment, were subjected to immunofluorescent staining for phospho-VE-cadherin. For the Piezo inhibitions, HUVEC were grown in transwell inserts and treated with TNF-α, followed by fluorescently (rhodamine B) labeled echogenic liposomes (RhB-ELIP). Concurrent treatments included Piezo 1 inhibitors (Jatrorrhizine and Tubeimoside I) and Piezo 2 inhibitor D-GsMTx4. Half the wells were treated with continuous wave US for 1 minute. After a 30-minute incubation at 37°C, lower well media fluorescence intensity was measured (544 Ex/590 Em) in a microplate reader. Results. US treatment of HUVEC caused increases in phospho-VE-cadherin expression, clustered near the periphery of cells, within 60 minutes (Fig. 1a), which was confirmed by Image J quantitation (Fig. 1b). No inhibition of US-enhanced RhB-ELIP passage through a HUVEC monolayer was seen in the presence of up to 10 µM Jatrorrhizine or Tubeimoside I, well within the effective range of the compounds (Fig. 1c), while 10 µM D-GsMTx4 completely inhibited US-enhanced passage (Fig. 1d), indicating that the effect is mediated by Piezo 2, but not Piezo 1. Conclusions. The results implicate mediation of US-enhanced permeability by Piezo 2, which mediates tactile responses rather than shear stress, and by VE-cadherin.
More abstracts on this topic:
Acute and Chronic Phosphorylation of CaMKII on Coronary Microvascular/Endothelial Function
Iddrisu Hanisah, Xing Hang, Shi Guangbin, Liu Yuhong, Feng Jun
Effects of pathogenic variation in ADAMTSL4 on vascular smooth muscle cell function relevant to spontaneous coronary artery dissectionPinnock Cameron, Hunker Kristina, Ganesh Santhi