Final ID: TU180
Pleiotropic and eQTL Analysis of Lipid Traits Identifies Novel Genetic Signals in African-Ancestry Populations
Abstract Body: Background: Dyslipidemia is a major, modifiable determinant of cardiovascular disease (CVD). However, the genetic architecture of lipid regulation remains incompletely understood, particularly in African-ancestry populations that experience a disproportionate burden of CVD but are underrepresented in genomic studies.
Methods: We conducted meta-analyses of genome-wide association summary statistics for HDL cholesterol, LDL cholesterol, triglycerides, and total cholesterol from the ACCME (n~8,800), AWI-Gen (11,700), and GRC-Uganda (n~6408) cohorts using METAL. Pleiotropic variants were identified with PLEIO, and genome-wide significant loci were assessed for tissue-specific regulatory activity using eQTpLot based on multi-tissue eQTL datasets.
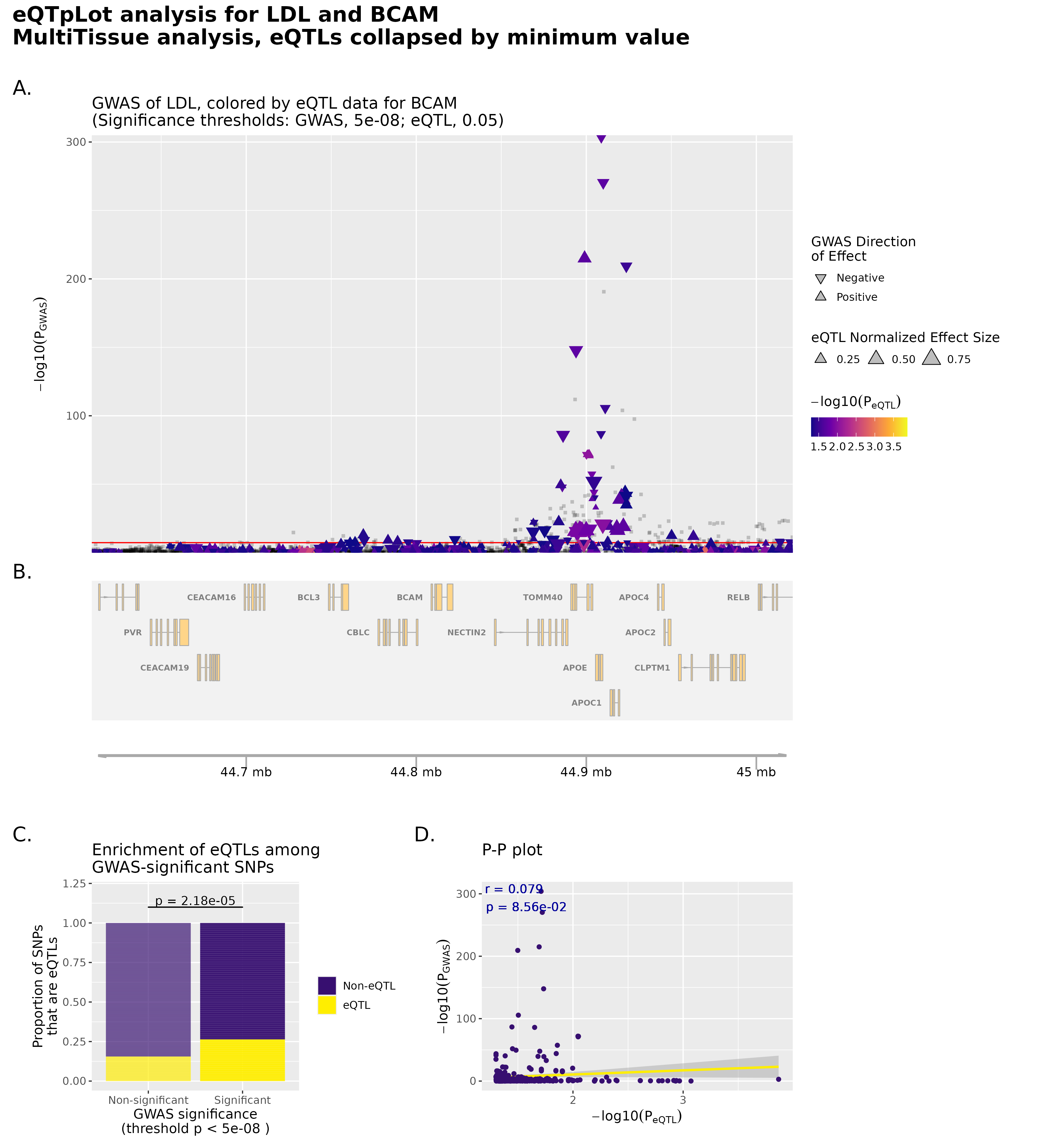
Results: We identified 90 genome-wide significant pleiotropic single nucleotide polymorphisms (SNPs) across eight chromosomes. The strongest associations occurred in the APOE, NECTIN2, TOMM40, and APOC1 region on chromosome 19, where the lead variant showed significant multi-trait effects across total cholesterol, LDL, and HDL. Additional genome-wide significant pleiotropic variants were detected near NECTIN2 (P = 8.09 x 10^-133), CETP (P = 9.44 x 10^-90), LIPC (P = 9.42 x 10^-51), PSRC1 (P = 6.99 x 10^-32), and LPL (P = 5.79 x 10^-22). Expression quantitative trait locus (eQTL) analyses demonstrated that these pleiotropic SNPs affected gene expression in lipid-related tissues, including BCAM and APOE in liver and adipose tissue and CETP in liver and arterial wall. These results highlight shared transcriptional regulation across multiple lipid traits within biologically established loci.
Conclusions: These findings reveal shared biological pathways underlying dyslipidemia and provide functional evidence linking genetic variation to gene expression in diverse populations. By characterizing ancestry-specific pleiotropic architecture, this work establishes a foundation for developing targeted therapeutic strategies and more accurate, ancestry-informed genomic tools for cardiovascular risk assessment and prevention.
Methods: We conducted meta-analyses of genome-wide association summary statistics for HDL cholesterol, LDL cholesterol, triglycerides, and total cholesterol from the ACCME (n~8,800), AWI-Gen (11,700), and GRC-Uganda (n~6408) cohorts using METAL. Pleiotropic variants were identified with PLEIO, and genome-wide significant loci were assessed for tissue-specific regulatory activity using eQTpLot based on multi-tissue eQTL datasets.
Results: We identified 90 genome-wide significant pleiotropic single nucleotide polymorphisms (SNPs) across eight chromosomes. The strongest associations occurred in the APOE, NECTIN2, TOMM40, and APOC1 region on chromosome 19, where the lead variant showed significant multi-trait effects across total cholesterol, LDL, and HDL. Additional genome-wide significant pleiotropic variants were detected near NECTIN2 (P = 8.09 x 10^-133), CETP (P = 9.44 x 10^-90), LIPC (P = 9.42 x 10^-51), PSRC1 (P = 6.99 x 10^-32), and LPL (P = 5.79 x 10^-22). Expression quantitative trait locus (eQTL) analyses demonstrated that these pleiotropic SNPs affected gene expression in lipid-related tissues, including BCAM and APOE in liver and adipose tissue and CETP in liver and arterial wall. These results highlight shared transcriptional regulation across multiple lipid traits within biologically established loci.
Conclusions: These findings reveal shared biological pathways underlying dyslipidemia and provide functional evidence linking genetic variation to gene expression in diverse populations. By characterizing ancestry-specific pleiotropic architecture, this work establishes a foundation for developing targeted therapeutic strategies and more accurate, ancestry-informed genomic tools for cardiovascular risk assessment and prevention.
More abstracts on this topic:
Associations between sleep quality and individual lipid abnormalities in Hong Kong Chinese women: Findings from the MECH-HK study
Chen Mingyu, Xie Yao Jie, Cheung Kin
Association of African Genetic Ancestry with Lipid Profile and Dyslipidemias in the Tobago Health StudyBeresford Ruel, Miljkovic Iva, Zmuda Joseph, Cvejkus Ryan, Wheeler Victor, Kuipers Allison