Final ID: WE403
Development and Implementation of an Artificial Intelligence-Based Predictive Model for Sudden Cardiac Death by Integrating Personal Health Records and Clinical Data
Abstract Body: Background:
In Japan, approximately 90,000 individuals die annually from sudden cardiac death (SCD). Predicting SCD using electronic health records (EHRs) from hospitals remains difficult. The growing use of wearable devices enables the continuous collection of personal health records (PHRs) and offers an opportunity to develop robust databases by integrating PHRs with EHRs. Applying artificial intelligence (AI) to these combined datasets may enable the early detection of SCD precursors.
Objective:
To develop an AI-based predictive model for SCD and related cardiovascular events by integrating PHRs with EHRs.
Methods:
We are prospectively enrolling high-risk patients—those with prior heart failure (HF), acute coronary syndrome (ACS), or out-of-hospital cardiac arrest—from seven Japanese centers. Participants use wearable devices (e.g., Fitbit, Apple Watch, or VINSTA ring) and home monitors to collect body weight and blood pressure. These data is integrated with EHRs, including clinical outcomes, laboratory tests, electrocardiograms, and echocardiograms. The primary endpoint is SCD, and the secondary endpoints include ACS, lethal arrhythmias, and hospitalization for HF. AI-based analysis was utilized to identify physiological changes preceding cardiovascular events.
Results:
From April 2024 to October 2025, 212 patients (mean age 58 ± 13 years; 77% male) were enrolled, yielding 167 person-years of follow-up. There were 12 cardiovascular events (3 HF, 3 ACS, 6 lethal arrhythmias) and no SCD. Temporary analysis revealed:
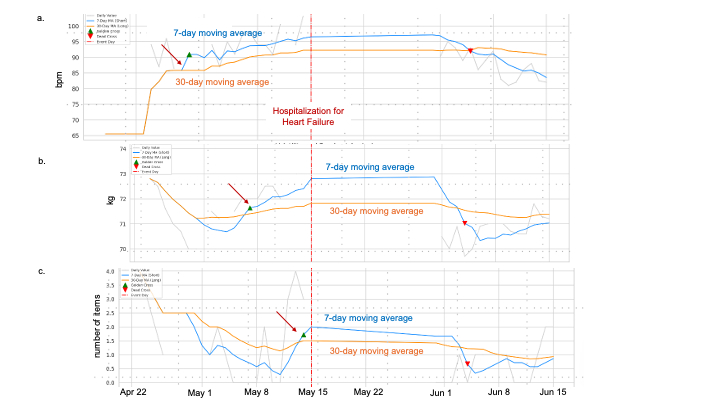
1. In HF, pulse rate increased from baseline 14 days prior, followed by weight gain seven days prior, and increased subjective symptoms three days before hospitalization. (Fig. 1)
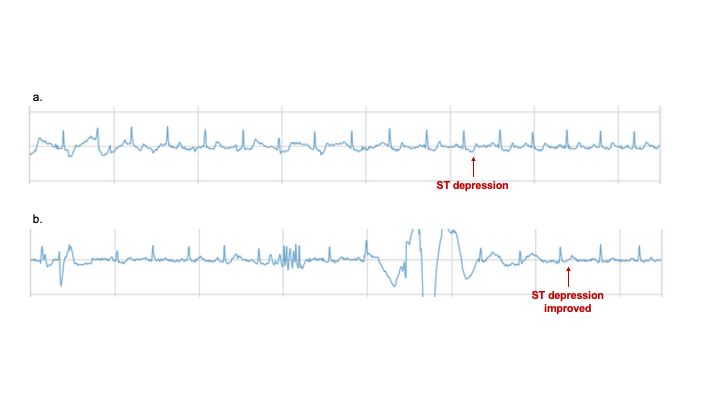
2. In ACS, ST-segment depression detected by VINSTA ring appeared two days before the onset and resolved after treatment. (Fig. 2)
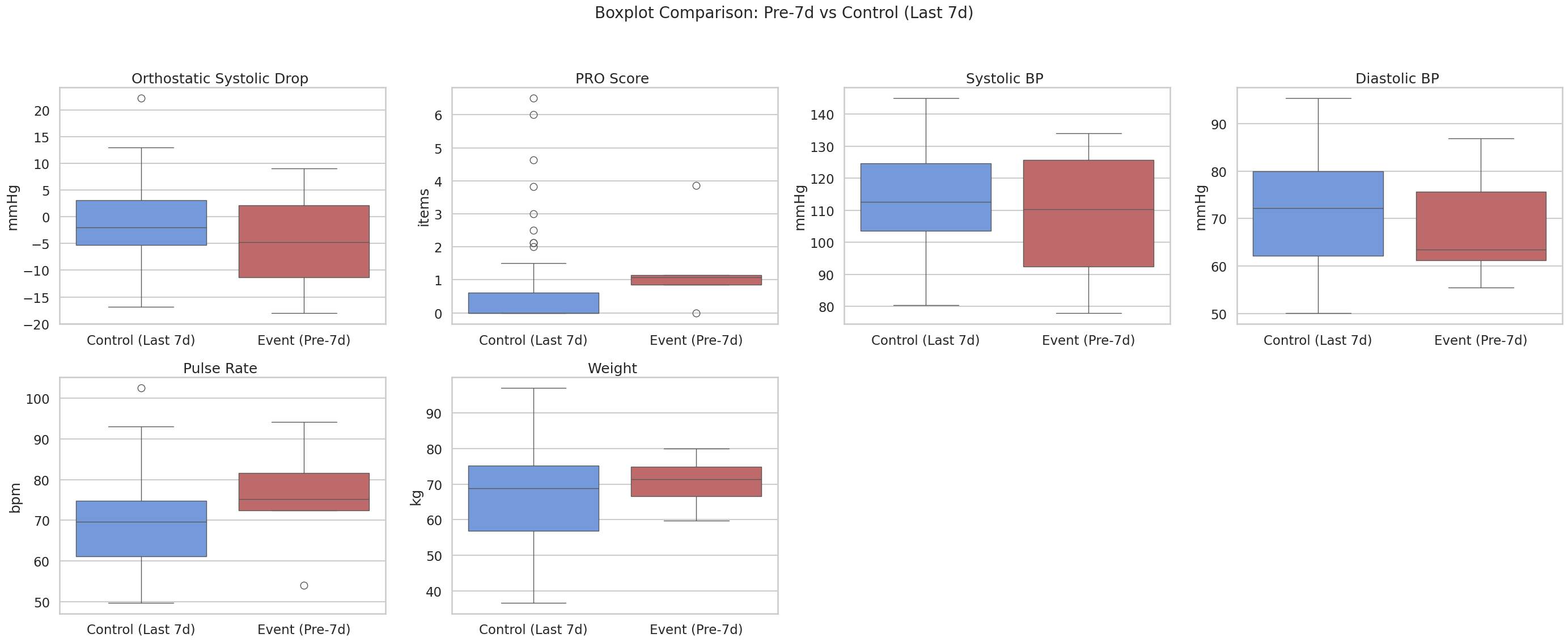
3. Event-positive participants showed higher mean pulse rate (81 ± 11 bpm vs. 70 ± 11 bpm; n = 4 vs. 66), greater orthostatic systolic drop (-2.3 ± 14.1 mmHg vs. -0.5 ± 7.2 mmHg; n = 2 vs. 19), and more frequent reports of subjective symptoms (1.9 ± 1.4 items vs. 0.7 ± 1.4 items; n = 4 vs. 57) seven days prior to the events. (Fig.3)
Discussion:
Integrating PHRs with EHRs enables continuous, personalized monitoring and early detection of cardiovascular deterioration. AI-driven analysis of these multimodal data may substantially enhance SCD risk prediction and support timely interventions to prevent adverse events.
In Japan, approximately 90,000 individuals die annually from sudden cardiac death (SCD). Predicting SCD using electronic health records (EHRs) from hospitals remains difficult. The growing use of wearable devices enables the continuous collection of personal health records (PHRs) and offers an opportunity to develop robust databases by integrating PHRs with EHRs. Applying artificial intelligence (AI) to these combined datasets may enable the early detection of SCD precursors.
Objective:
To develop an AI-based predictive model for SCD and related cardiovascular events by integrating PHRs with EHRs.
Methods:
We are prospectively enrolling high-risk patients—those with prior heart failure (HF), acute coronary syndrome (ACS), or out-of-hospital cardiac arrest—from seven Japanese centers. Participants use wearable devices (e.g., Fitbit, Apple Watch, or VINSTA ring) and home monitors to collect body weight and blood pressure. These data is integrated with EHRs, including clinical outcomes, laboratory tests, electrocardiograms, and echocardiograms. The primary endpoint is SCD, and the secondary endpoints include ACS, lethal arrhythmias, and hospitalization for HF. AI-based analysis was utilized to identify physiological changes preceding cardiovascular events.
Results:
From April 2024 to October 2025, 212 patients (mean age 58 ± 13 years; 77% male) were enrolled, yielding 167 person-years of follow-up. There were 12 cardiovascular events (3 HF, 3 ACS, 6 lethal arrhythmias) and no SCD. Temporary analysis revealed:
1. In HF, pulse rate increased from baseline 14 days prior, followed by weight gain seven days prior, and increased subjective symptoms three days before hospitalization. (Fig. 1)
2. In ACS, ST-segment depression detected by VINSTA ring appeared two days before the onset and resolved after treatment. (Fig. 2)
3. Event-positive participants showed higher mean pulse rate (81 ± 11 bpm vs. 70 ± 11 bpm; n = 4 vs. 66), greater orthostatic systolic drop (-2.3 ± 14.1 mmHg vs. -0.5 ± 7.2 mmHg; n = 2 vs. 19), and more frequent reports of subjective symptoms (1.9 ± 1.4 items vs. 0.7 ± 1.4 items; n = 4 vs. 57) seven days prior to the events. (Fig.3)
Discussion:
Integrating PHRs with EHRs enables continuous, personalized monitoring and early detection of cardiovascular deterioration. AI-driven analysis of these multimodal data may substantially enhance SCD risk prediction and support timely interventions to prevent adverse events.
More abstracts on this topic:
AI-Driven Gait Classification for Peripheral Artery Disease (PAD) Detection Using Machine Learning and Nonlinear Gait Dynamics
Mohammadzadeh Gonabadi Arash, Fallahtafti Farahnaz, Pipinos Iraklis, Burnfield Judith, Myers Sara
Automated Personalized Modulation of the Sympathetic “fight-or-flight” Reflex In Vivo Using Implantable Microdevices for Enhancing Neurocardiac Function and Reducing Mortality Risk.Kundu Dibyasankha, Hazra Debapriya, Mukherjee Shayani, Jones Steven, Dey Swati, Demazumder Deeptankar