Final ID: WE465
Pharmacologic Treatment To Lower Blood Pressure In Hypertensive Pregnant Women Living In Low- And Middle-Income Countries: Systematic Review
Abstract Body: Background: Hypertension (HTN) is a leading cause of maternal mortality, especially in low- and middle-income countries (LMICs) where >90% of HTN-related deaths occur. HTN control during pregnancy is essential for improved maternal and neonatal outcomes.
Methods: A systematic literature search was conducted to identify trials of pharmacologic blood pressure (BP) lowering in pregnant women with peripartum HTN living in LMICs. PubMed, Embase, Cochrane Library, Global Index Medicus, and the WHO International Clinical Trials Registry Platform were searched from inception to December 6, 2024. Two investigators independently screened and extracted data. Primary outcome was difference in mean arterial pressure (MAP) change between trial arms.
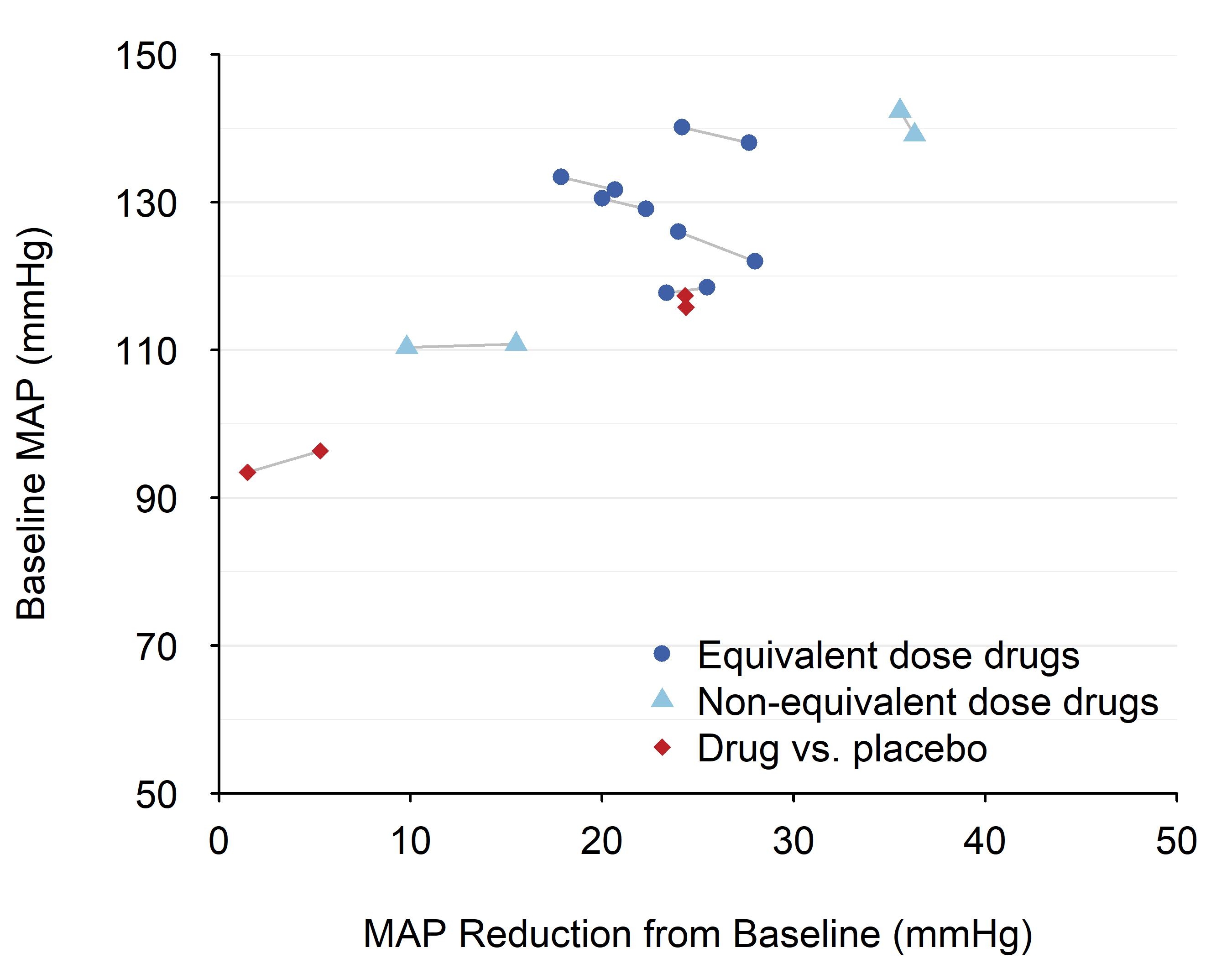
Results: A total of 2272 unique articles were identified; 2252 were excluded during screening and 9 during full-text review. Included were 11 trials with a total of 2118 pregnant females living in Africa or Asia; sample size ranged from 70 to 894. Drugs studied included nifedipine, labetalol, hydralazine, or placebo. Nine compared either equivalent or non-equivalent dose strengths of two antihypertensive drugs; two compared drug to placebo. Difference in MAP between treatment groups was reported by or calculated for nine trials, ranging from 0.07 to 5.7 mmHg; there was no consistent pattern of greater BP reduction according to trial design (equivalent or non-equivalent dose strength or drug vs placebo; Figure). Neither was there a consistent pattern for between group systolic or diastolic BP difference. Oral nifedipine and labetalol led to fewer non-serious side effects compared with hydralazine or methyldopa. No differences were observed in serious adverse event rates.
Conclusion: A systematic review found only 11 trials of pharmacologic BP lowering in pregnant women with HTN living in LMICs, highlighting need for more high-quality trials in this population. No consistent difference in BP-lowering efficacy between drugs was observed. Our results suggest tolerability, ease of administration, cost, and availability are likely more important factors to guide selection of antihypertensive drug treatment of HTN in pregnant women living in LMICs.
Methods: A systematic literature search was conducted to identify trials of pharmacologic blood pressure (BP) lowering in pregnant women with peripartum HTN living in LMICs. PubMed, Embase, Cochrane Library, Global Index Medicus, and the WHO International Clinical Trials Registry Platform were searched from inception to December 6, 2024. Two investigators independently screened and extracted data. Primary outcome was difference in mean arterial pressure (MAP) change between trial arms.
Results: A total of 2272 unique articles were identified; 2252 were excluded during screening and 9 during full-text review. Included were 11 trials with a total of 2118 pregnant females living in Africa or Asia; sample size ranged from 70 to 894. Drugs studied included nifedipine, labetalol, hydralazine, or placebo. Nine compared either equivalent or non-equivalent dose strengths of two antihypertensive drugs; two compared drug to placebo. Difference in MAP between treatment groups was reported by or calculated for nine trials, ranging from 0.07 to 5.7 mmHg; there was no consistent pattern of greater BP reduction according to trial design (equivalent or non-equivalent dose strength or drug vs placebo; Figure). Neither was there a consistent pattern for between group systolic or diastolic BP difference. Oral nifedipine and labetalol led to fewer non-serious side effects compared with hydralazine or methyldopa. No differences were observed in serious adverse event rates.
Conclusion: A systematic review found only 11 trials of pharmacologic BP lowering in pregnant women with HTN living in LMICs, highlighting need for more high-quality trials in this population. No consistent difference in BP-lowering efficacy between drugs was observed. Our results suggest tolerability, ease of administration, cost, and availability are likely more important factors to guide selection of antihypertensive drug treatment of HTN in pregnant women living in LMICs.
More abstracts on this topic:
Application of Angiotensin-Based Biomarkers Quantified by Mass Spectrometry to Predict Preeclampsia
Shoemaker Robin, Poglitsch Marko, Huang Hong, Mirsky Elizabeth, Cockerham Cynthia, Bauer John, O'brien John
A Randomized Comparison of Online Motivational Themes in Cardiovascular Clinical Trial RecruitmentHussain Zaib, Harry Tamunotonye, Michos Erin, Milller Hailey, Juraschek Stephen, Turkson-ocran Ruth-alma, Lahey Timothy, Feng Yuanyuan, Plante Timothy