Final ID: MP11
Association of fixed dose combination therapy use with blood pressure control and cardiovascular outcomes in the Systolic Blood Pressure Reduction Intervention (SPRINT) Trial: A post-hoc analysis
Abstract Body: Background: The seminal SPRINT trial demonstrated that intensive blood pressure (BP) control is associated with lower risk of adverse cardiovascular (CV) outcomes. However, BP control among individuals with hypertension in the US remains suboptimal. Fixed-dose combination (FDC) therapies are commonly used in clinical practice as a strategy to improve adherence to BP-lowering therapies. Whether use of FDC agents can facilitate greater intensive BP control and lower risk of adverse CV outcomes is not known.
Methods: We performed a post-hoc analysis of SPRINT including 8623 participants with available drug therapy data. FDC use was defined by use of at least one combination agent. The association between FDC use and rate of change in BP in the acute (≤6 months) follow-up of the trial was assessed by linear mixed-effect model with repeated measures (LMMRM) including a FDC*time interaction term with adjustment for potential confounders. Difference in adjusted estimated marginal means for follow-up BP achieved at different timepoints between the FDC vs. non-FDC groups was also compared using LMMRM. Finally, multivariable Cox proportional hazards models were constructed to evaluate the association of FDC use with risk of composite CV events (non-fatal MI, CV death, and HF event).
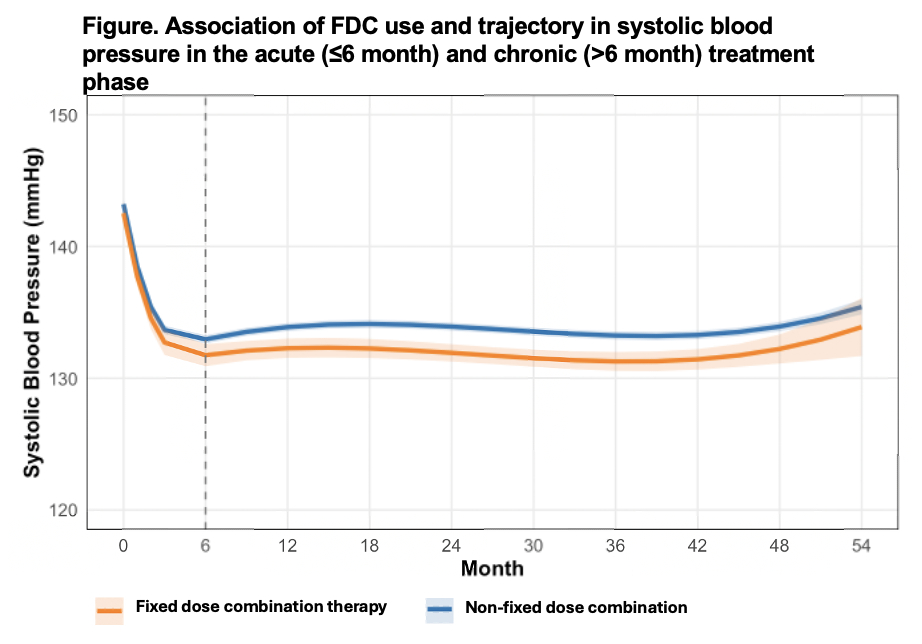
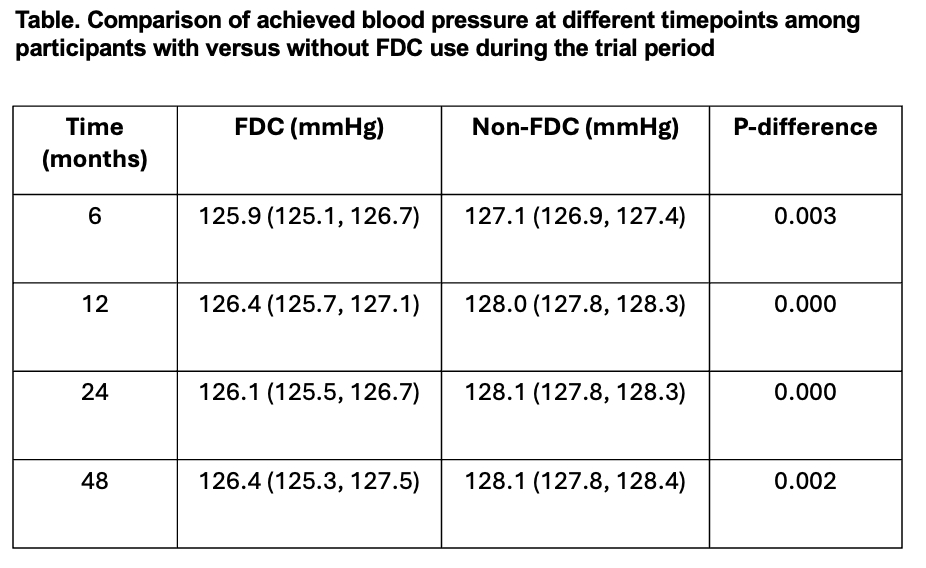
Results: Among 8623 participants analyzed, 9.3% (N=803) were prescribed FDC at baseline with greater rate of use in the intensive BP control vs. usual care group (5.79[5.61 to 5.97] vs. 3.90[3.75 to 4.06] per 100 person-months; p-diff<0.001). Among those with FDC use, the most commonly used formulations were lisinopril/HCTZ (11.3%) and triamterene/HCTZ (9.1%). In the overall cohort, FDC use was associated with more rapid BP reduction in the first 6 months (mean monthly change in systolic BP FDC vs. non-FDC [95% CI]: -1.94[-1.65 to -1.22] mmHg vs. -1.33[-1.27 to -1.40] mmHg; p-diff<0.001) with comparable difference across treatment arms. On long-term follow-up, participants using FDCs achieved significantly lower systolic BP at each timepoint. The risk of the composite CV events was not different among those with vs. without FDC use during the trial after adjusting for potential confounders (HR[95%CI]: 0.81[0.49 to 1.34]).
Conclusions: Among SPRINT participants, use of FDC therapies was associated with more rapid acute BP control and lower BP during long-term follow-up. These observations suggest that FDC therapies may facilitate faster and more sustained achievement of intensive BP control.
Methods: We performed a post-hoc analysis of SPRINT including 8623 participants with available drug therapy data. FDC use was defined by use of at least one combination agent. The association between FDC use and rate of change in BP in the acute (≤6 months) follow-up of the trial was assessed by linear mixed-effect model with repeated measures (LMMRM) including a FDC*time interaction term with adjustment for potential confounders. Difference in adjusted estimated marginal means for follow-up BP achieved at different timepoints between the FDC vs. non-FDC groups was also compared using LMMRM. Finally, multivariable Cox proportional hazards models were constructed to evaluate the association of FDC use with risk of composite CV events (non-fatal MI, CV death, and HF event).
Results: Among 8623 participants analyzed, 9.3% (N=803) were prescribed FDC at baseline with greater rate of use in the intensive BP control vs. usual care group (5.79[5.61 to 5.97] vs. 3.90[3.75 to 4.06] per 100 person-months; p-diff<0.001). Among those with FDC use, the most commonly used formulations were lisinopril/HCTZ (11.3%) and triamterene/HCTZ (9.1%). In the overall cohort, FDC use was associated with more rapid BP reduction in the first 6 months (mean monthly change in systolic BP FDC vs. non-FDC [95% CI]: -1.94[-1.65 to -1.22] mmHg vs. -1.33[-1.27 to -1.40] mmHg; p-diff<0.001) with comparable difference across treatment arms. On long-term follow-up, participants using FDCs achieved significantly lower systolic BP at each timepoint. The risk of the composite CV events was not different among those with vs. without FDC use during the trial after adjusting for potential confounders (HR[95%CI]: 0.81[0.49 to 1.34]).
Conclusions: Among SPRINT participants, use of FDC therapies was associated with more rapid acute BP control and lower BP during long-term follow-up. These observations suggest that FDC therapies may facilitate faster and more sustained achievement of intensive BP control.
More abstracts on this topic:
A Selective Agonist of the Beta 2 Retinoic Acid Receptor Reduces Necrosis, Lipid Peroxidation, and Oxidative Stress in Doxorubicin-induced Cardiotoxicity
Varzideh Fahimeh, Jankauskas Stanislovas, Kansakar Urna, Tang Xiao-han, Gudas Lorraine, Levi Roberto, Gambardella Jessica, Santulli Gaetano
APOL1 Risk Variants in Preeclampsia: From Mechanistic Insights to Therapeutic OpportunitiesRao Vivikta, Waikar Sushrut, Wei Jin, Fu Jiayi, Liaw Easton, De Thea, Sedarski Jonathan, Tang Janice, Zhang Jie, Kuohung Wendy, Ilori Titilayo