Final ID: Wed008
A soluble guanylate cyclase stimulator improves survival in a rat model of heart failure with reduced ejection fraction and chronic kidney disease induced by aorto-caval fistula and 5/6 nephrectomy
Abstract Body: Background
New therapeutic strategies for heart failure (HF) combined with chronic kidney disease (CKD) are needed, and the current knowledge suggests that targeting the NO/sGC/cGMP pathway could be a promising approach.
Purpose
We hypothesised that chronic soluble guanylate cyclase (sGC) stimulation would attenuate the course of rodent HFrEF induced by volume overload due to aorto-caval fistula (ACF) combined with CKD caused by 5/6 nephrectomy (5/6 Nx).
Methods
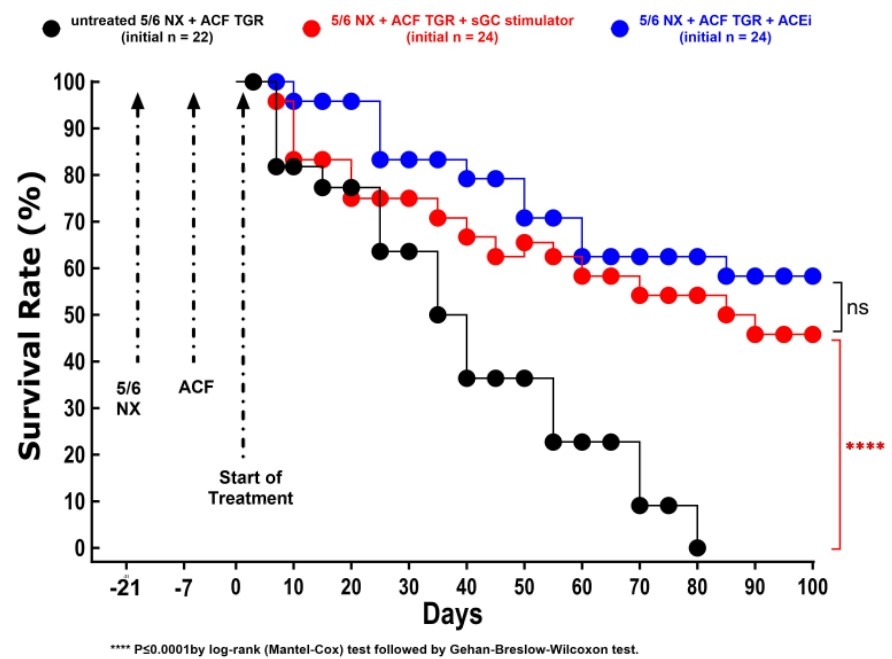
5/6 Nx was performed in Ren-2 transgenic rats (TGR), a model of angiotensin II-dependent hypertension, at the animal age of 8 weeks. One week later, ACF was created; another two weeks later, the animals were randomly divided into three groups, and the therapy was started: sGC stimulation with BAY-41-8543 (sGC stim.) (3 mg.kg-1.day-1 in food, n = 24), angiotensin-converting enzyme inhibitor (ACEi) (trandolapril, 2 mg.L-1 in drinking water, n = 24), or placebo (n = 22). Sham-operated TGR represented a control group (n = 8). The follow-up period was 20 weeks. GraphPad Prism 10 was used for statistical analysis with a log-rank (Mantel-Cox) test to analyse survival data.
Results
All sham-operated rats survived till the end of the study. On the contrary, all untreated HF+CKD rats died by week 12. The treatment with the sGC stim. or with ACEi similarly improved the survival rate: 46 % in sGC stim. group (p < 0,0001 vs. placebo) and 58 % in the ACEi group (p < 0,0001 vs. placebo) (Figure 1).
Conclusion(s)
Our results indicate that in the ACF-5/6Nx TGR animal model of combined heart failure (HF) and chronic kidney disease (CKD), treatment with an sGC stimulator reduces mortality to a similar extent as ACE inhibitor (ACEi) monotherapy compared to placebo. Further preclinical research is needed to better understand the NO/sGC/cGMP pathway in HF with concurrent CKD.
New therapeutic strategies for heart failure (HF) combined with chronic kidney disease (CKD) are needed, and the current knowledge suggests that targeting the NO/sGC/cGMP pathway could be a promising approach.
Purpose
We hypothesised that chronic soluble guanylate cyclase (sGC) stimulation would attenuate the course of rodent HFrEF induced by volume overload due to aorto-caval fistula (ACF) combined with CKD caused by 5/6 nephrectomy (5/6 Nx).
Methods
5/6 Nx was performed in Ren-2 transgenic rats (TGR), a model of angiotensin II-dependent hypertension, at the animal age of 8 weeks. One week later, ACF was created; another two weeks later, the animals were randomly divided into three groups, and the therapy was started: sGC stimulation with BAY-41-8543 (sGC stim.) (3 mg.kg-1.day-1 in food, n = 24), angiotensin-converting enzyme inhibitor (ACEi) (trandolapril, 2 mg.L-1 in drinking water, n = 24), or placebo (n = 22). Sham-operated TGR represented a control group (n = 8). The follow-up period was 20 weeks. GraphPad Prism 10 was used for statistical analysis with a log-rank (Mantel-Cox) test to analyse survival data.
Results
All sham-operated rats survived till the end of the study. On the contrary, all untreated HF+CKD rats died by week 12. The treatment with the sGC stim. or with ACEi similarly improved the survival rate: 46 % in sGC stim. group (p < 0,0001 vs. placebo) and 58 % in the ACEi group (p < 0,0001 vs. placebo) (Figure 1).
Conclusion(s)
Our results indicate that in the ACF-5/6Nx TGR animal model of combined heart failure (HF) and chronic kidney disease (CKD), treatment with an sGC stimulator reduces mortality to a similar extent as ACE inhibitor (ACEi) monotherapy compared to placebo. Further preclinical research is needed to better understand the NO/sGC/cGMP pathway in HF with concurrent CKD.
More abstracts on this topic:
ANSWER-HF: A Randomized Controlled Trial of Sacubitril/Valsartan Versus Enalapril in Patients with Chronic Chagas Cardiomyopathy and Reduced Ejection Fraction
Madrini Junior Vagner, Antunes Talita, Damiani Lucas, Jose Grupi Cesar, Mathias Junior Wilson, Lopes Renato, Ramires Felix, Ramos Souza Paulo, Fernandes Fabio, Maria Ianni Barbara, Silva Martins Alan, JadÁn Luzuriaga Georgina Del Cisne, Cardoso Barbosa Fonseca Keila, Ribeiro Orlando, Bispo Da Cruz Allecineia
Acculturation and Cardiovascular-Kidney-Metabolic Syndrome: a Study of Immigrant Adults From the National Health and Nutrition Examination SurveyChakrabarti Amit, Le Austin, Elfassy Tali, Yang Eugene