Final ID: Su4004
Spatial Organizations of Heterochromatin Underlie Cardiomyocyte Nuclear Structural Integrity Against Mechanical Stress
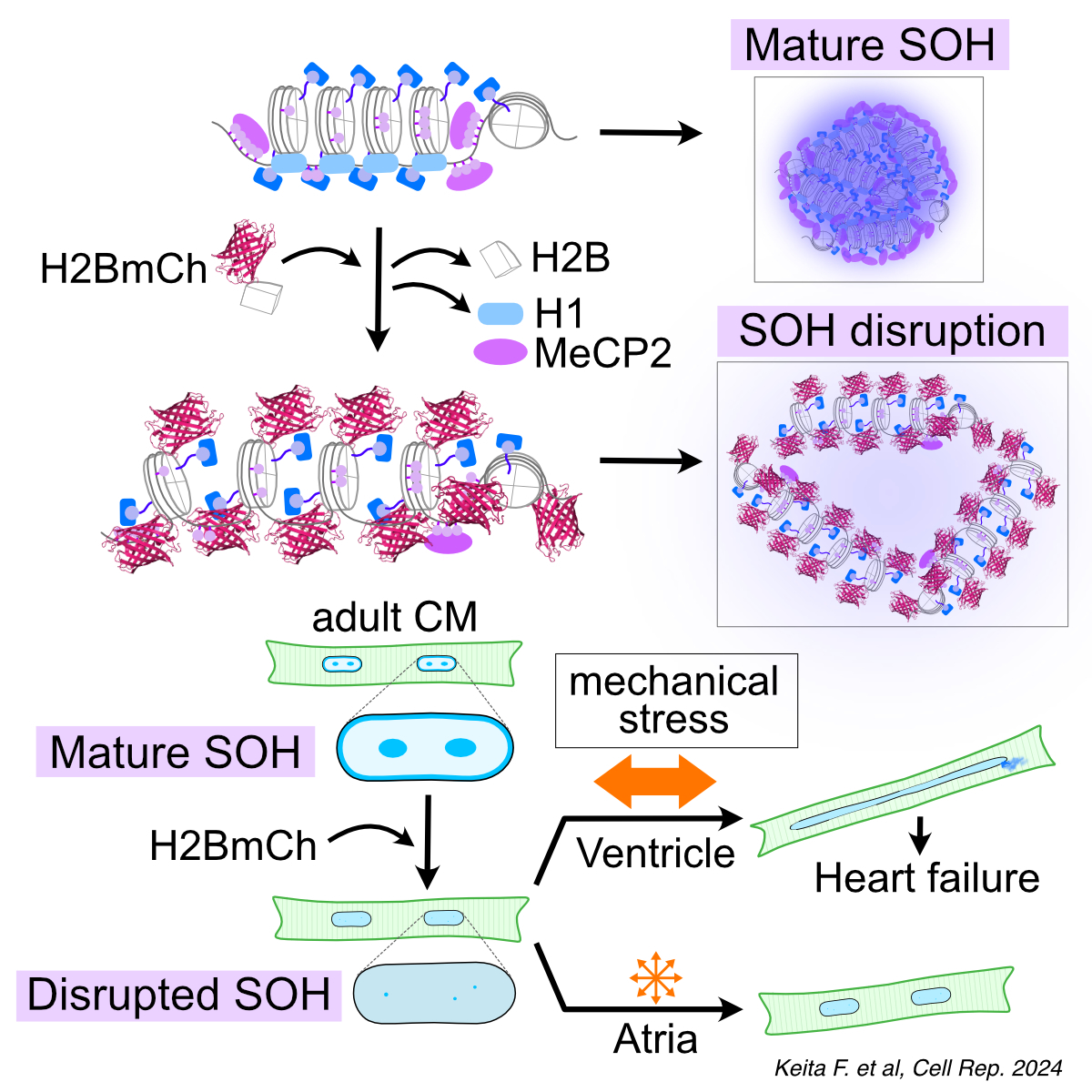
Abstract Body (Do not enter title and authors here): Introduction: Cardiomyocyte (CM) nuclei endure relentless contraction cycles while preserving nuclear architecture and transcriptional fidelity. In mice engineered for ErbB4-driven CM proliferation—with H2B-mCherry as a CM nuclei marker—we unexpectedly observed heterochromatin loss, extreme nuclear elongation, envelope rupture, and fatal heart failure, implicating a physical role for heterochromatin in nuclear integrity.
Hypothesis: We propose that spatial organizations of heterochromatin (SOH)—a peripheral layer plus chromocenters visible by microscopy—serve as a structural platform essential for CM nuclear integrity.
Methods: We generated mice with CM-specific overexpression (OE) of H2B-mCherry, H2B alone, or NLS-mCherry using Troponin T–Cre (constitutive) and MerCreMer (inducible). Cardiac function was monitored by echocardiography. Nuclear morphology was examined by confocal and electron microscopy; stiffness by atomic force microscopy. Hi-C and ATAC-seq profiled chromatin topology and accessibility; RNA-seq assessed transcription. Western blots quantified histone composition. Statistics: two-tailed t-tests or ANOVA (α=0.05).
Results: Constitutive H2B-mCherry OE—but not H2B or NLS-mCherry—induced progressive CM nuclear elongation and lethal cardiomyopathy. In adult-inducible OE, SOH disappeared within two weeks–preceding any change in nuclear shape. CM nuclei then softened (a ~50% drop in Young’s modulus from 3.2→1.6kPa, p<0.001), elongated over two-fold, and ~23% ruptured, releasing DNA that activated cGAS/STING (cGAS ↑18.3-fold, p<0.01), driving inflammation and fibrosis. Hi-C revealed blurred TAD insulation, while ATAC-seq/RNA-seq were unchanged, indicating that SOH disruption compromises mechanics rather than gene expression. Westerns demonstrated H2B-mCherry incorporation displaces endogenous H2B and, via mCherry steric hindrance, dislodges histone H1 (–60%, p<0.001), loosening chromatin compaction, and disperses LLPS factors (e.g., MeCP2), abolishing SOH. Aged hearts recapitulated SOH dissipation, H1 reduction, and nuclear deformation, linking heterochromatin disruption to age-related cardiomyopathies.
Conclusion: Our data identify SOH as critical mechanical support for CM nuclei; their disruption softens nuclei, precipitates deformation and inflammation, and culminates in heart failure, highlighting heterochromatin maintenance as a novel therapeutic target in age-related cardiomyopathies.
Hypothesis: We propose that spatial organizations of heterochromatin (SOH)—a peripheral layer plus chromocenters visible by microscopy—serve as a structural platform essential for CM nuclear integrity.
Methods: We generated mice with CM-specific overexpression (OE) of H2B-mCherry, H2B alone, or NLS-mCherry using Troponin T–Cre (constitutive) and MerCreMer (inducible). Cardiac function was monitored by echocardiography. Nuclear morphology was examined by confocal and electron microscopy; stiffness by atomic force microscopy. Hi-C and ATAC-seq profiled chromatin topology and accessibility; RNA-seq assessed transcription. Western blots quantified histone composition. Statistics: two-tailed t-tests or ANOVA (α=0.05).
Results: Constitutive H2B-mCherry OE—but not H2B or NLS-mCherry—induced progressive CM nuclear elongation and lethal cardiomyopathy. In adult-inducible OE, SOH disappeared within two weeks–preceding any change in nuclear shape. CM nuclei then softened (a ~50% drop in Young’s modulus from 3.2→1.6kPa, p<0.001), elongated over two-fold, and ~23% ruptured, releasing DNA that activated cGAS/STING (cGAS ↑18.3-fold, p<0.01), driving inflammation and fibrosis. Hi-C revealed blurred TAD insulation, while ATAC-seq/RNA-seq were unchanged, indicating that SOH disruption compromises mechanics rather than gene expression. Westerns demonstrated H2B-mCherry incorporation displaces endogenous H2B and, via mCherry steric hindrance, dislodges histone H1 (–60%, p<0.001), loosening chromatin compaction, and disperses LLPS factors (e.g., MeCP2), abolishing SOH. Aged hearts recapitulated SOH dissipation, H1 reduction, and nuclear deformation, linking heterochromatin disruption to age-related cardiomyopathies.
Conclusion: Our data identify SOH as critical mechanical support for CM nuclei; their disruption softens nuclei, precipitates deformation and inflammation, and culminates in heart failure, highlighting heterochromatin maintenance as a novel therapeutic target in age-related cardiomyopathies.
More abstracts on this topic:
ALOX15 mediates thrombin-induced myristoylation, trafficking and interaction of PARs leading to platelet activation and hemostasis
Govatati Suresh, Hoque Md Monirul, Thota Swapna, Rao Gadiparthi
A Bridge from Sweet to Sour: A Case of Recurrent Myocardial Stunning in Diabetic KetoacidosisSatish Vikyath, Pargaonkar Sumant, Slipczuk Leandro, Schenone Aldo, Maliha Maisha, Chi Kuan Yu, Sunil Kumar Sriram, Borkowski Pawel, Vyas Rhea, Rodriguez Szaszdi David Jose Javier, Kharawala Amrin, Seo Jiyoung