Final ID: MP2779
Mavacamten monotherapy in real-world patients with obstructive hypertrophic cardiomyopathy: Evidence from COLLIGO-HCM
Abstract Body (Do not enter title and authors here): Background: The mavaCamten ObservationaL evIdence Global cOnsortium in hypertrophic cardiomyopathy (COLLIGO-HCM, ClinicalTrials.gov ID NCT06372457) is a global observational study describing real-world outcomes of obstructive HCM patients treated with mavacamten. Limited real-world evidence exists regarding the use of mavacamten as monotherapy or following reduction or withdrawal of standard of care background therapies such as beta-blockers or calcium channel blockers.
Purpose: To describe the real-world outcomes of patients treated with mavacamten monotherapy.
Methods: This retrospective study uses patient-level data from existing medical records and electronic registries from COLLIGO-HCM sites in the US, Canada, the UK, Australia, and Israel. Patient characteristics, NYHA class, echocardiography data, and safety were analyzed at baseline and follow-up visits, up to week 36 in four cohorts: monotherapy group (n=88), defined as those with monotherapy at initiation (n=20) plus monotherapy after discontinuation of background therapy (n=68), mavacamten in combination with background therapy that has been reduced (n=13), mavacamten in combination with stable, unchanged background therapy over the duration of the study period (n=177), and the overall cohort (n= 278).
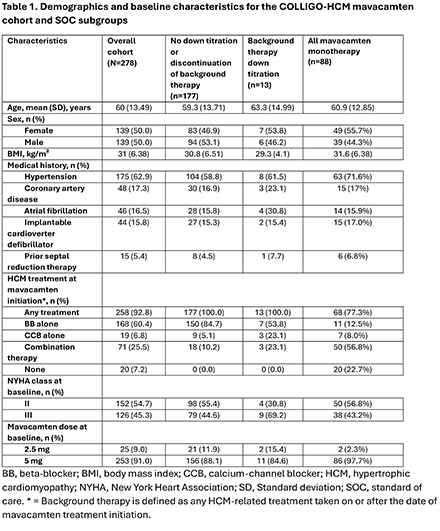
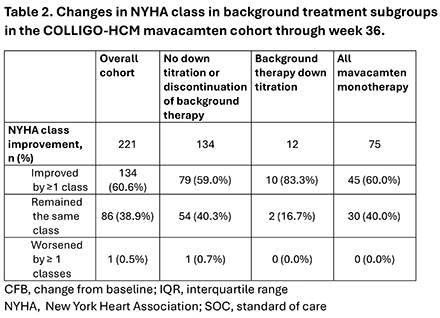
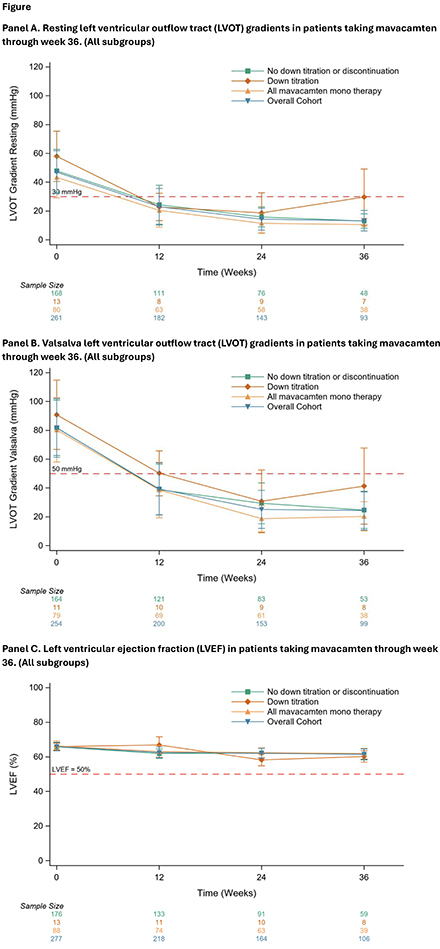
Results: The mavacamten monotherapy cohort included 88/278 patients (31.7%). Baseline clinical and demographic characteristics include: 55.7% female, 56.8% NYHA Class II and 43.2% Class III, high BMI (mean: 31.6 kg/m2), history of atrial fibrillation (15.9%) and hypertension (71.6%). The median follow-up time was 35.9 weeks (IQR:21.9,116.9) (Table 1). By week 36, 60% of patients achieved ≥ 1 NYHA class improvement. Additionally, mean left ventricular outflow tract (LVOT) gradients of ≤30 mm Hg at rest and with Valsalva were achieved in 97.3% and 81.6% of patients, respectively (Table 2. Figure). Mean left ventricular ejection fraction (LVEF) post mavacamten initiation remained at or above 62% throughout follow-up (baseline value 66.4%). Two patients in the monotherapy cohort permanently discontinued mavacamten due to LVEF<50%, both recovered after discontinuing treatment.
Conclusions: This analysis demonstrates effectiveness, safety and consistency of mavacamten monotherapy with previously reported monotherapy analyses from pivotal, long-term and real-world studies.
Purpose: To describe the real-world outcomes of patients treated with mavacamten monotherapy.
Methods: This retrospective study uses patient-level data from existing medical records and electronic registries from COLLIGO-HCM sites in the US, Canada, the UK, Australia, and Israel. Patient characteristics, NYHA class, echocardiography data, and safety were analyzed at baseline and follow-up visits, up to week 36 in four cohorts: monotherapy group (n=88), defined as those with monotherapy at initiation (n=20) plus monotherapy after discontinuation of background therapy (n=68), mavacamten in combination with background therapy that has been reduced (n=13), mavacamten in combination with stable, unchanged background therapy over the duration of the study period (n=177), and the overall cohort (n= 278).
Results: The mavacamten monotherapy cohort included 88/278 patients (31.7%). Baseline clinical and demographic characteristics include: 55.7% female, 56.8% NYHA Class II and 43.2% Class III, high BMI (mean: 31.6 kg/m2), history of atrial fibrillation (15.9%) and hypertension (71.6%). The median follow-up time was 35.9 weeks (IQR:21.9,116.9) (Table 1). By week 36, 60% of patients achieved ≥ 1 NYHA class improvement. Additionally, mean left ventricular outflow tract (LVOT) gradients of ≤30 mm Hg at rest and with Valsalva were achieved in 97.3% and 81.6% of patients, respectively (Table 2. Figure). Mean left ventricular ejection fraction (LVEF) post mavacamten initiation remained at or above 62% throughout follow-up (baseline value 66.4%). Two patients in the monotherapy cohort permanently discontinued mavacamten due to LVEF<50%, both recovered after discontinuing treatment.
Conclusions: This analysis demonstrates effectiveness, safety and consistency of mavacamten monotherapy with previously reported monotherapy analyses from pivotal, long-term and real-world studies.
More abstracts on this topic:
Acute Severe Mitral Regurgitation Due to Flail Posterior Leaflet without Chordal Rupture Following Myosin Inhibitor Treatment of Hypertrophic Obstructive Cardiomyopathy
Patel Shreyan, Taha Israa, Elmi Daniel, Shirani Jamshid
AI-enhanced Electrocardiographic Evaluation of Left Ventricular Ejection Fraction and Outflow Tract Gradient in Hypertrophic CardiomyopathySangha Veer, Aminorroaya Arya, Dhingra Lovedeep, Pedroso Aline, Oikonomou Evangelos, Khera Rohan