Final ID: Su3003
Body weight reduction-associated and -unassociated changes in lipid profile in people with obesity, with or without T2D, treated with tirzepatide: post-hoc analysis of SURMOUNT-1 and SURMOUNT-2
Abstract Body (Do not enter title and authors here): Background: Tirzepatide is a once weekly GIP and GLP-1 receptor agonist approved for the treatment of type 2 diabetes (T2D) and obesity. This post hoc analysis evaluated the contribution of body weight reduction-associated and -unassociated effects on the lipid profile of tirzepatide-treated participants living with obesity, without and with T2D, from the SURMOUNT-1 and SURMOUNT-2 clinical trials, respectively.
Methods: Participants treated with tirzepatide (pooled doses of 5, 10, 15 mg in SURMOUNT-1 [N=1765] and 10 and 15 mg in SURMOUNT-2 [N=587]) were included in this analysis. The estimated treatment effects and the body weight reduction-associated and -unassociated attribution on changes from baseline in lipid profile (total cholesterol, HDL-C, LDL-C, non-HDL-C, VLDL-C, and triglycerides) at 24 and 72 weeks were assessed via the SAS CAUSALMED procedure.
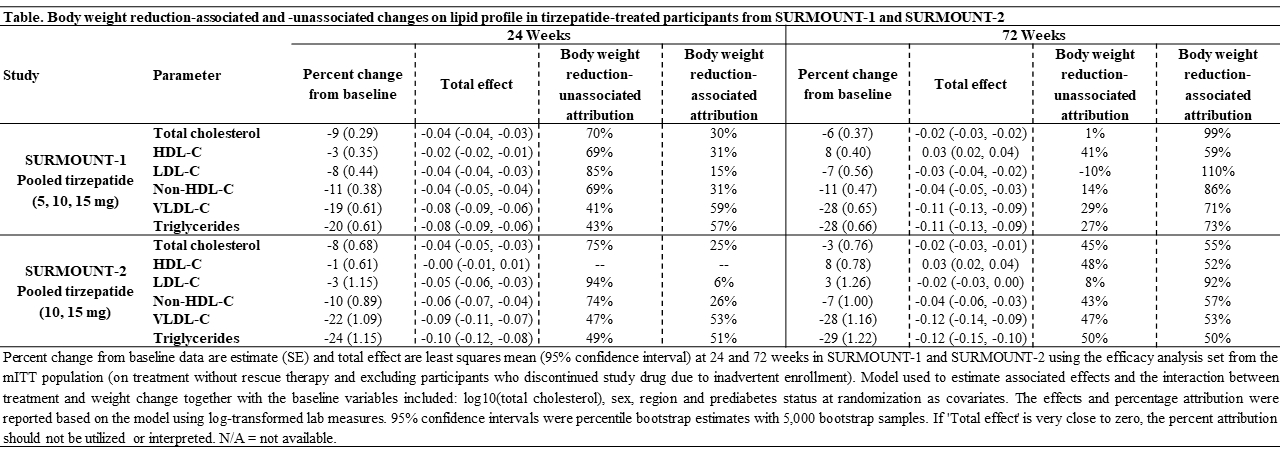
Results: In SURMOUNT-1, after 24 weeks of tirzepatide treatment in participants without T2D, 69-85% of the changes in total cholesterol, HDL-C, LDL-C, and non-HDL-C, and 41-43% of the changes in VLDL-C and triglycerides occurred unassociated with body weight reduction (Table). At 72 weeks, most of the effect on the lipid profile was associated with body weight reduction. In SURMOUNT-2, after 24 weeks of tirzepatide treatment in participants with T2D, most of the changes in the lipid profile occurred unassociated with body weight reduction. At 72 weeks, changes observed in the lipid profile were predominantly associated with body weight reduction, although 43-50% of those changes also occurred unassociated with body weight reduction, except for LDL-C which was almost completely (92%) associated with body weight reduction.
Conclusions: In this post hoc analysis from SURMOUNT-1 and SURMOUNT-2, changes in lipid profile were mostly unassociated with body weight reduction after 24 weeks of tirzepatide treatment and associated with of body weight reduction at 72 weeks. Body weight reduction-unassociated mechanisms responsible for the initial changes in lipid profile in participants with obesity treated with tirzepatide warrants further research studies.
Methods: Participants treated with tirzepatide (pooled doses of 5, 10, 15 mg in SURMOUNT-1 [N=1765] and 10 and 15 mg in SURMOUNT-2 [N=587]) were included in this analysis. The estimated treatment effects and the body weight reduction-associated and -unassociated attribution on changes from baseline in lipid profile (total cholesterol, HDL-C, LDL-C, non-HDL-C, VLDL-C, and triglycerides) at 24 and 72 weeks were assessed via the SAS CAUSALMED procedure.
Results: In SURMOUNT-1, after 24 weeks of tirzepatide treatment in participants without T2D, 69-85% of the changes in total cholesterol, HDL-C, LDL-C, and non-HDL-C, and 41-43% of the changes in VLDL-C and triglycerides occurred unassociated with body weight reduction (Table). At 72 weeks, most of the effect on the lipid profile was associated with body weight reduction. In SURMOUNT-2, after 24 weeks of tirzepatide treatment in participants with T2D, most of the changes in the lipid profile occurred unassociated with body weight reduction. At 72 weeks, changes observed in the lipid profile were predominantly associated with body weight reduction, although 43-50% of those changes also occurred unassociated with body weight reduction, except for LDL-C which was almost completely (92%) associated with body weight reduction.
Conclusions: In this post hoc analysis from SURMOUNT-1 and SURMOUNT-2, changes in lipid profile were mostly unassociated with body weight reduction after 24 weeks of tirzepatide treatment and associated with of body weight reduction at 72 weeks. Body weight reduction-unassociated mechanisms responsible for the initial changes in lipid profile in participants with obesity treated with tirzepatide warrants further research studies.
More abstracts on this topic:
Intrinsic Motivation for Engaging in Healthy Lifestyle Behaviors is Associated with Reduced Postpartum Weight Retention
Brown Susan, Ferrara Assiamira, Kiernan Michaela, Hedderson Monique, Quesenberry Charles, Smith Bridgette, Millman Andrea, Serrato Bandera Hillary, Daredia Saher, Shan Jun
Association between Elevated Lipoprotein(a) and Cardiovascular Events and Mortality in a Nationally Representative Sample of US Medicare, Medicaid, and Commercial Enrollees with ASCVDHu Xingdi, Lozama Tony, Petrilla Allison, Agatep Barnabie, Mcmorrow Donna, Mohammadi Iman, Reisman Lonny, Wong Nathan