Final ID: Wed095
Extracelluar Matrix Proteins Regulate Endothelial Activation and Priming During Atherosclerosis
Abstract Body: Atherosclerosis is a chronic inflammatory condition that affects large arteries, characterized by the recruitment of circulating leukocytes to clear accumulated lipids from vessel walls. At atherosclerosis-prone sites, the endothelial lining becomes activated, expressing adhesion molecules and altering its basement membrane composition by depositing fibronectin. Endothelial cells exposed to fibronectin exhibit heightened inflammatory activation, promoting plaque formation.
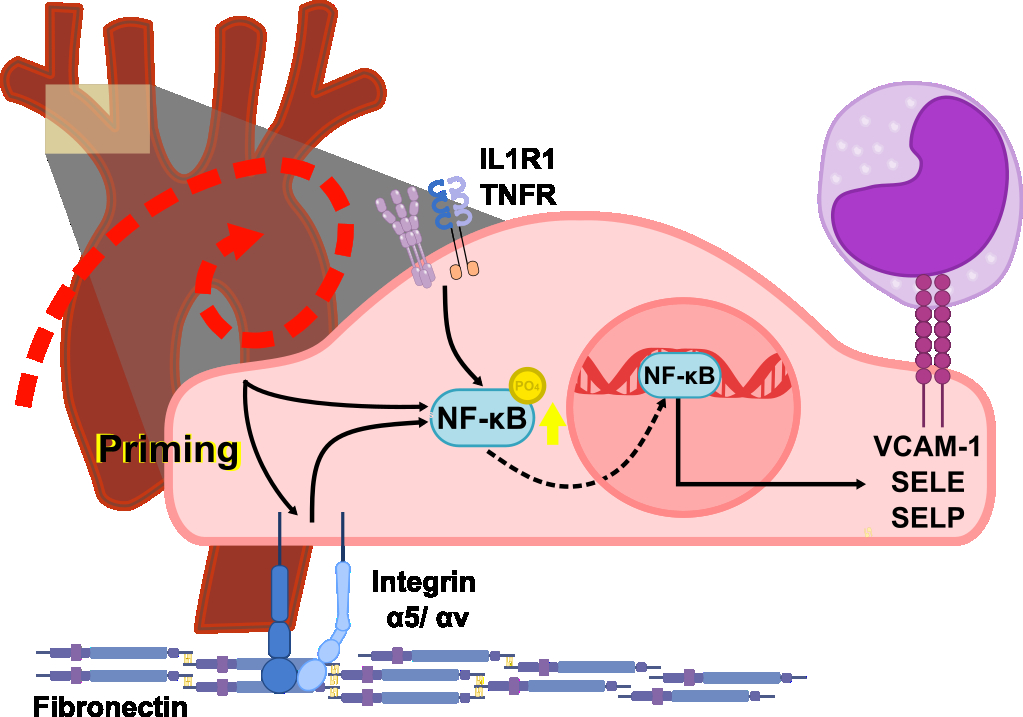
Studies have shown that endothelial cells in atheroprone areas become primed. Endothelial priming involves an increase in the expression of components of inflammatory pathways, which enhances the strength of their response to inflammatory stimuli. It’s been shown that reducing fibronectin deposition in the vascular wall helps mitigate the formation of plaque in mouse models, though the underlying mechanisms remain unclear. Deleting either of the fibronectin binding integrins, α5 or αv, in endothelial cells significantly reduces plaque formation in mouse models. RNAseq data from our lab, taken from integrin α5/αv double knockout human aortic endothelial cells, show a decrease in cytokine-cytokine receptor interaction genes. Prior experiments show that the inhibition of these integrins blunts NFκB-mediated inflammatory responses to weaker pro-atherogenic stimuli, like flow and oxidized LDL, but not to cytokines like IL-1β and TNFα. Based on this, we hypothesized that simultaneous deletion of both integrins would reduce inflammatory response inflammatory cytokines on endothelial cells and impair advanced plaque formation.
To test this hypothesis, we used mouse models with inducible, endothelial-specific double knockout of integrins α5 and αv, alongside in vitro studies using knockout mouse lung endothelial cells and siRNA knockdown in human aortic endothelial cells treated with IL-1β and TNFα. Deletion of α5 alone or with αv prevented plaque formation in a 16-week high-fat diet model, but only combined deletion halted progression of existing plaques. In vitro, integrin deletion reduced endothelial responses to cytokines, lowering ICAM-1 and VCAM-1 expression at both protein and mRNA levels. Despite no changes in upstream signaling events, NFκB total protein levels were reduced, suggesting a priming effect. Collectively, these findings indicate that integrins α5 and αv contribute to atherosclerosis by sensitizing endothelial cells to inflammatory cytokines through NFκB expression regulation.
Studies have shown that endothelial cells in atheroprone areas become primed. Endothelial priming involves an increase in the expression of components of inflammatory pathways, which enhances the strength of their response to inflammatory stimuli. It’s been shown that reducing fibronectin deposition in the vascular wall helps mitigate the formation of plaque in mouse models, though the underlying mechanisms remain unclear. Deleting either of the fibronectin binding integrins, α5 or αv, in endothelial cells significantly reduces plaque formation in mouse models. RNAseq data from our lab, taken from integrin α5/αv double knockout human aortic endothelial cells, show a decrease in cytokine-cytokine receptor interaction genes. Prior experiments show that the inhibition of these integrins blunts NFκB-mediated inflammatory responses to weaker pro-atherogenic stimuli, like flow and oxidized LDL, but not to cytokines like IL-1β and TNFα. Based on this, we hypothesized that simultaneous deletion of both integrins would reduce inflammatory response inflammatory cytokines on endothelial cells and impair advanced plaque formation.
To test this hypothesis, we used mouse models with inducible, endothelial-specific double knockout of integrins α5 and αv, alongside in vitro studies using knockout mouse lung endothelial cells and siRNA knockdown in human aortic endothelial cells treated with IL-1β and TNFα. Deletion of α5 alone or with αv prevented plaque formation in a 16-week high-fat diet model, but only combined deletion halted progression of existing plaques. In vitro, integrin deletion reduced endothelial responses to cytokines, lowering ICAM-1 and VCAM-1 expression at both protein and mRNA levels. Despite no changes in upstream signaling events, NFκB total protein levels were reduced, suggesting a priming effect. Collectively, these findings indicate that integrins α5 and αv contribute to atherosclerosis by sensitizing endothelial cells to inflammatory cytokines through NFκB expression regulation.
More abstracts on this topic:
A Loss of Function Polymorphism in the Propeptide of Lysyl Oxidase Exacerbates Atherosclerosis
Jung In-hyuk, Amrute Junedh, Luna Sophia, Wagoner Ryan, Lee Paul, Burks Kendall, Holloway Karyn, Alisio Arturo, Stitziel Nathan
An Unexpected MYOCD-Driven Constraint on VSMC Adaptive States Promotes Aortic AneurysmIshimwe Nestor, Wu Xiaoliang, Shi Hong, Miano Joseph, Yan Chen, Long Xiaochun