Final ID: 60
Effect of Multivitamin Reduces Within-Visit Blood Pressure Variability in Older Adults: Findings from the COSMOS Randomized Controlled Trial
Abstract Body: Background: Within-Visit Blood Pressure Variability (WVBPV) reflects short-term fluctuations in BP readings. Although less studied than mean blood pressure, WVBPV is an independent predictor of cardiovascular events and mortality. The effect of multivitamin-multimineral (MVM) supplementation on WVBPV is unknown. We examined the two-year impact of MVM use on WVBPV in the COcoa Supplement and Multivitamin Outcomes Study (COSMOS).
Methods: COSMOS is a randomized, double-blind, placebo-controlled, 2×2 factorial trial testing daily MVM (Centrum Silver) and cocoa extract (CE) supplements for 2 years in women aged ≥65 years and men aged ≥60 years without major cardiovascular diseases (CVD) or recent cancer. In the COSMOS-Clinic subcohort with clinic-based measurements, WVBPV was assessed at baseline and 2 years follow-up using variability independent of the mean (VIM) and average real variability (ARV) of systolic BP (SBP). Treatment effects were estimated with mixed-effects models, adjusting for CE, age, sex, and recruitment source.
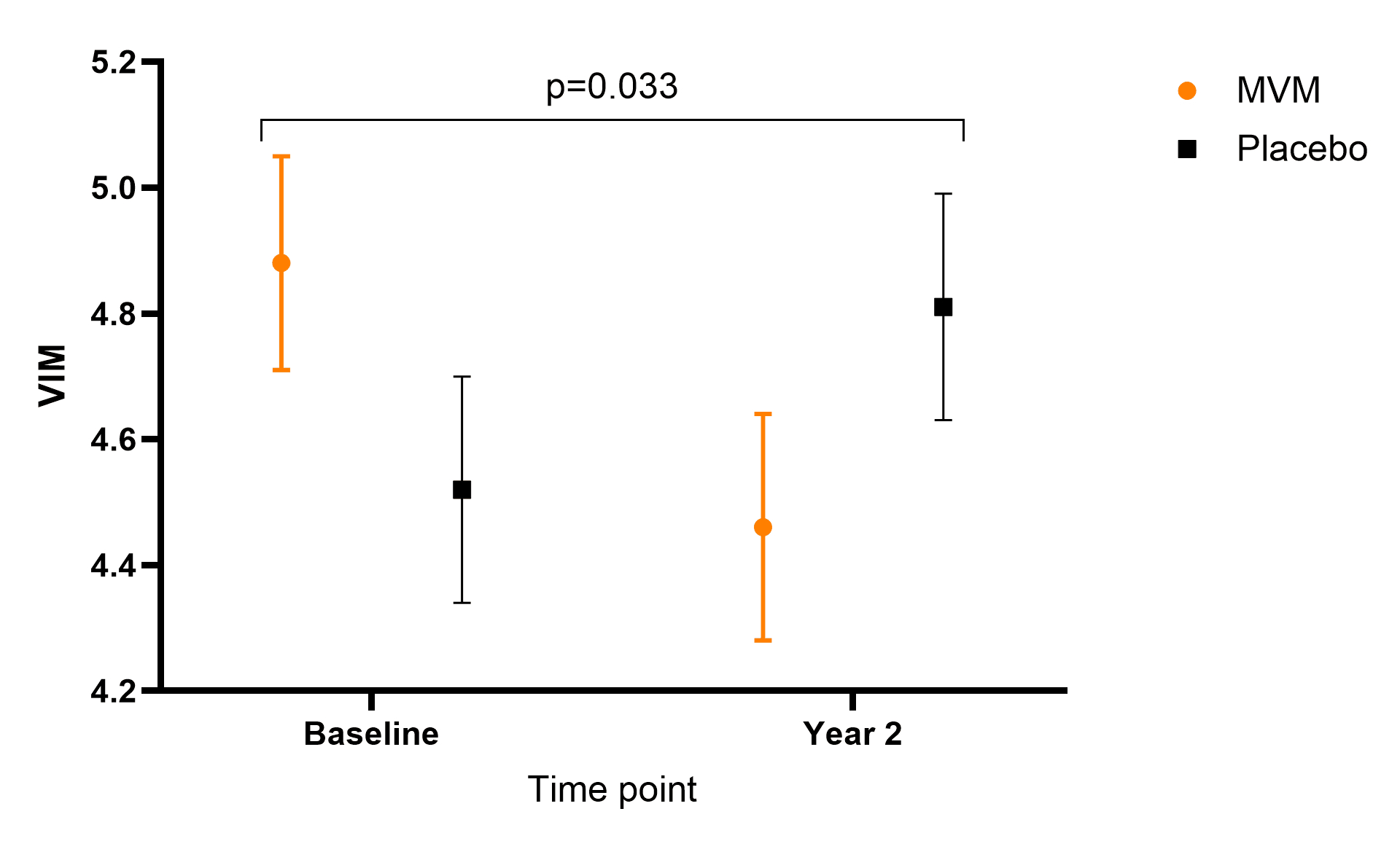
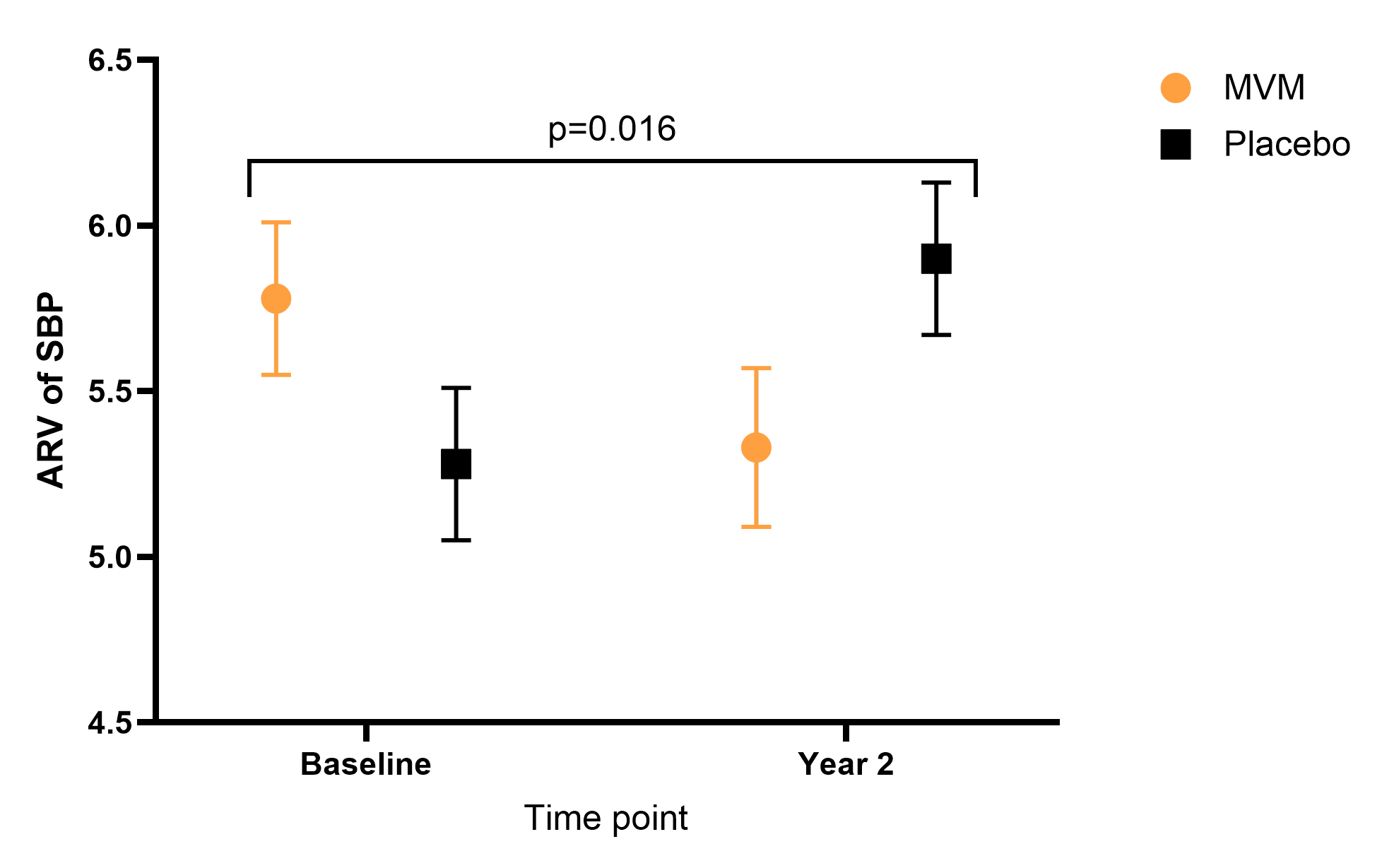
Results: Among 529 participants (mean age 69.7 ± 5.4 years; 48.8% women), baseline characteristics were similar by MVM treatment group. MVM supplementation reduced both VIM (−0.72; 95% CI: −1.38 to −0.06; p = 0.033) and ARV (−1.07 mmHg; 95% CI: −1.94 to −0.20; p = 0.016) over two years, without changing mean BPs. Reductions in ARV were greater females compared with men (−2.14 mmHg; 95% CI −3.35 to −0.92; p_interaction = 0.018).
Conclusions: This study provides the first randomized trial evidence that daily MVM supplementation improves WVBPV in older adults. Benefits appear to be more pronounced in women. Given the strong association between WVBPV and cardiovascular risk, these findings suggest a potential novel role for MVM in cardiovascular prevention strategies, warranting further evaluation of its clinical relevance.
Registration: URL: https://clinicaltrials.gov unique identifier NCT02422745
Methods: COSMOS is a randomized, double-blind, placebo-controlled, 2×2 factorial trial testing daily MVM (Centrum Silver) and cocoa extract (CE) supplements for 2 years in women aged ≥65 years and men aged ≥60 years without major cardiovascular diseases (CVD) or recent cancer. In the COSMOS-Clinic subcohort with clinic-based measurements, WVBPV was assessed at baseline and 2 years follow-up using variability independent of the mean (VIM) and average real variability (ARV) of systolic BP (SBP). Treatment effects were estimated with mixed-effects models, adjusting for CE, age, sex, and recruitment source.
Results: Among 529 participants (mean age 69.7 ± 5.4 years; 48.8% women), baseline characteristics were similar by MVM treatment group. MVM supplementation reduced both VIM (−0.72; 95% CI: −1.38 to −0.06; p = 0.033) and ARV (−1.07 mmHg; 95% CI: −1.94 to −0.20; p = 0.016) over two years, without changing mean BPs. Reductions in ARV were greater females compared with men (−2.14 mmHg; 95% CI −3.35 to −0.92; p_interaction = 0.018).
Conclusions: This study provides the first randomized trial evidence that daily MVM supplementation improves WVBPV in older adults. Benefits appear to be more pronounced in women. Given the strong association between WVBPV and cardiovascular risk, these findings suggest a potential novel role for MVM in cardiovascular prevention strategies, warranting further evaluation of its clinical relevance.
Registration: URL: https://clinicaltrials.gov unique identifier NCT02422745
More abstracts on this topic:
Apabetalone Protects Against Heart Failure with Preserved Ejection Fraction by Suppressing Myocardial Inflammation
Costantino Sarah, Nazha Hamdani, Paneni Francesco, Gorica Era, Mohammed Shafeeq, Telesca Marialucia, Mongelli Alessia, Masciovecchio Valeria, Herwig Melissa, Ambrosini Samuele, Ruschitzka Frank
Clinical Study on The Blood Pressure Changes in Patients with VEGFR-TKI-induced Hypertension During Anti-tumor TreatmentLi Jiaxin, Yu Jing