Final ID: MP1103
The Klotho Protein Reduces Vascular Calcification via Suppressing GPX4-mediated Ferroptosis in Vascular Smooth Muscle Cells
Abstract Body (Do not enter title and authors here): OBJECTIVES
Chronic kidney disease (CKD) patients exhibit elevated cardiovascular risk linked to vascular calcification—pathological calcium-phosphate deposition impairing vascular compliance. Previous studies identified reduced α-Klotho expression in CKD, potentially exacerbating phosphate dyshomeostasis. Hyperphosphatemia promotes vascular calcification by inducing ferroptosis in vascular smooth muscle cells (VSMCs) via GSH/GPX4 pathway suppression. This study aimed to elucidate α-Klotho's role in regulating ferroptosis-dependent calcification.
METHODS
VSMCs were treated with high-phosphate (2.6 mM) medium to mimic CKD calcific stress, with/without recombinant α-Klotho (100 ng/ml). Osteogenic markers (Runx2, OST, ALP) were assessed by western blot. Ferroptosis markers (Ptgs2/COX2, GSH, GPX4) and lipid peroxidation (Liperfluo) were analyzed via RT-qPCR/western blot. Intracellular Fe2+ (FerroOrange) and viability (CCK-8) were quantified. Ferroptosis inducer RSL3 was used for validation.
RESULTS
(1) High-phosphate-induced calcification & ferroptosis were attenuated by α-Klotho:
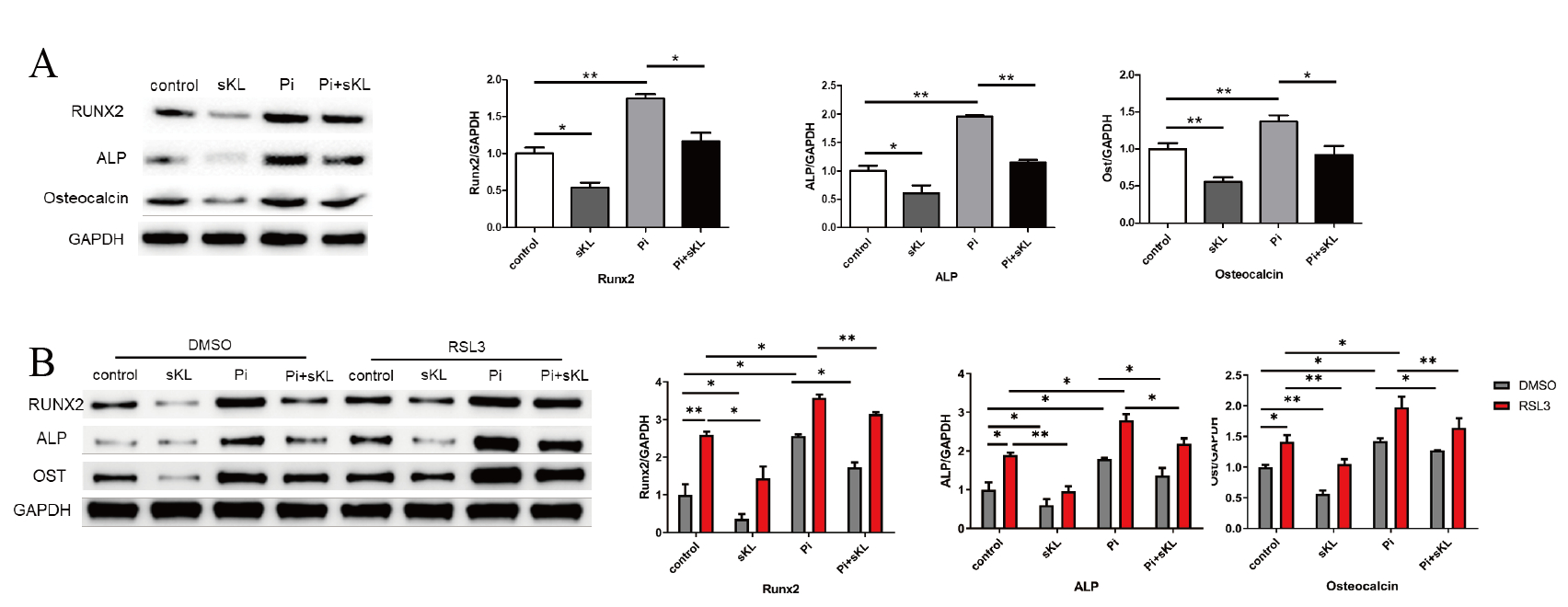
(i) High-phosphate significantly increased osteogenic markers (Runx2, ALP, OCN; P<0.05 vs. control), suppressed by α-Klotho (Fig 1A).
(ii) α-Klotho inhibited RSL3-induced VSMC calcification (Fig 1B).
(2) α-Klotho restored redox homeostasis:
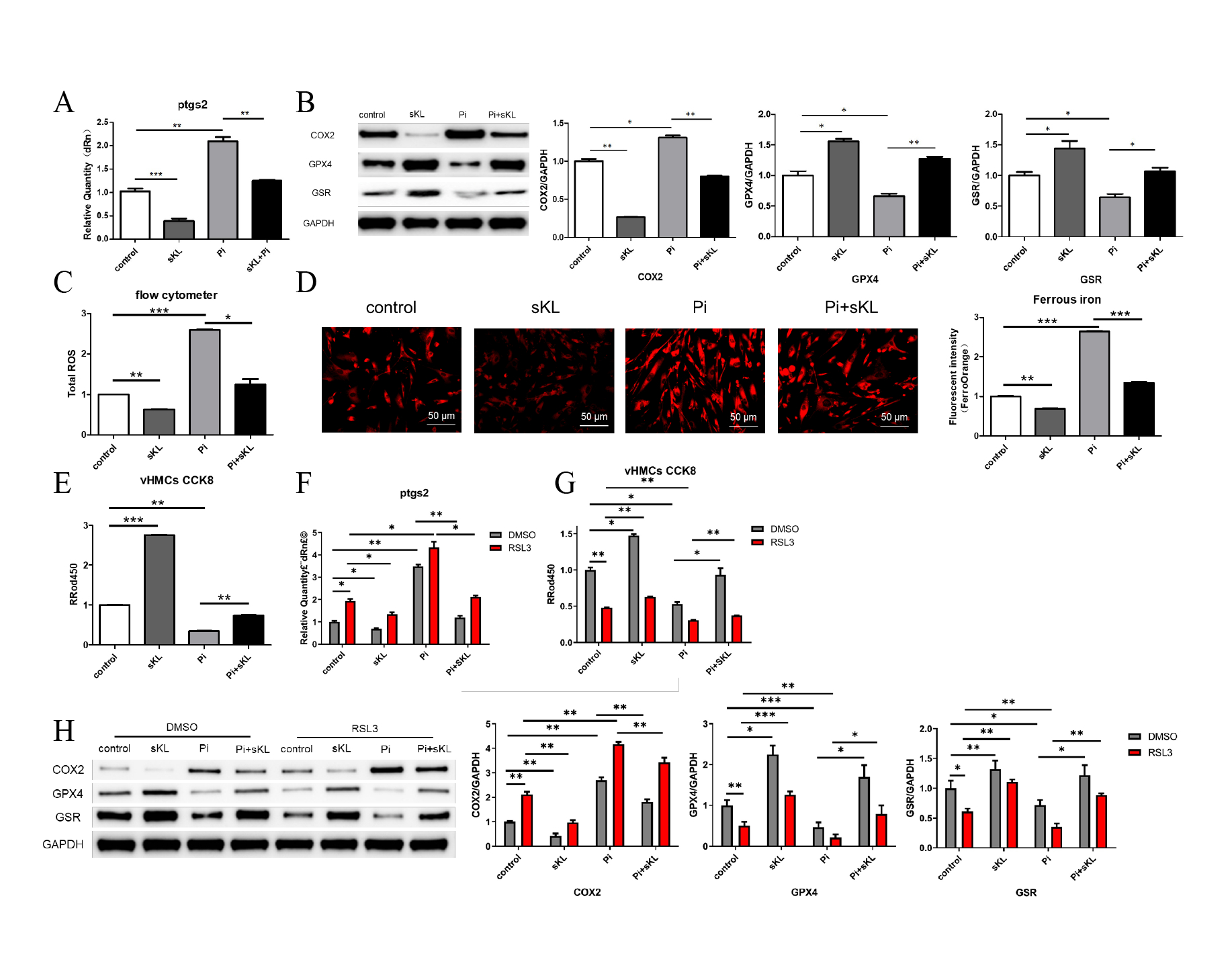
(i) High-phosphate upregulated COX2 (P<0.05) and downregulated GSH/GPX4; α-Klotho reversed these effects (Fig 2A-B).
(ii) High-phosphate increased lipid ROS and Fe2+ accumulation while reducing viability; α-Klotho suppressed lipid ROS/Fe2+ and restored viability (Fig 2C-E).
(iii) α-Klotho blocked RSL3-triggered calcification (Fig 2F-H).
Conclusion: α-Klotho inhibits High-phosphate-driven calcification via dual mechanisms: 1) Reactivating GSH/GPX4 to suppress ferroptosis; 2) Inhibiting VSMC osteogenic transdifferentiation.
CONCLUSIONS
α-Klotho inhibits High-phosphate-induced VSMC ferroptosis by activating GSH/GPX4, reducing lipid ROS/Fe2+ accumulation, and restoring viability. Concurrently, it downregulates osteogenic markers (Runx2/ALP/OCN), preventing phenotypic transition. These findings reveal α-Klotho's dual protection against vascular calcification via coordinated suppression of ferroptosis and osteogenic differentiation (Fig 1-2).
Chronic kidney disease (CKD) patients exhibit elevated cardiovascular risk linked to vascular calcification—pathological calcium-phosphate deposition impairing vascular compliance. Previous studies identified reduced α-Klotho expression in CKD, potentially exacerbating phosphate dyshomeostasis. Hyperphosphatemia promotes vascular calcification by inducing ferroptosis in vascular smooth muscle cells (VSMCs) via GSH/GPX4 pathway suppression. This study aimed to elucidate α-Klotho's role in regulating ferroptosis-dependent calcification.
METHODS
VSMCs were treated with high-phosphate (2.6 mM) medium to mimic CKD calcific stress, with/without recombinant α-Klotho (100 ng/ml). Osteogenic markers (Runx2, OST, ALP) were assessed by western blot. Ferroptosis markers (Ptgs2/COX2, GSH, GPX4) and lipid peroxidation (Liperfluo) were analyzed via RT-qPCR/western blot. Intracellular Fe2+ (FerroOrange) and viability (CCK-8) were quantified. Ferroptosis inducer RSL3 was used for validation.
RESULTS
(1) High-phosphate-induced calcification & ferroptosis were attenuated by α-Klotho:
(i) High-phosphate significantly increased osteogenic markers (Runx2, ALP, OCN; P<0.05 vs. control), suppressed by α-Klotho (Fig 1A).
(ii) α-Klotho inhibited RSL3-induced VSMC calcification (Fig 1B).
(2) α-Klotho restored redox homeostasis:
(i) High-phosphate upregulated COX2 (P<0.05) and downregulated GSH/GPX4; α-Klotho reversed these effects (Fig 2A-B).
(ii) High-phosphate increased lipid ROS and Fe2+ accumulation while reducing viability; α-Klotho suppressed lipid ROS/Fe2+ and restored viability (Fig 2C-E).
(iii) α-Klotho blocked RSL3-triggered calcification (Fig 2F-H).
Conclusion: α-Klotho inhibits High-phosphate-driven calcification via dual mechanisms: 1) Reactivating GSH/GPX4 to suppress ferroptosis; 2) Inhibiting VSMC osteogenic transdifferentiation.
CONCLUSIONS
α-Klotho inhibits High-phosphate-induced VSMC ferroptosis by activating GSH/GPX4, reducing lipid ROS/Fe2+ accumulation, and restoring viability. Concurrently, it downregulates osteogenic markers (Runx2/ALP/OCN), preventing phenotypic transition. These findings reveal α-Klotho's dual protection against vascular calcification via coordinated suppression of ferroptosis and osteogenic differentiation (Fig 1-2).
More abstracts on this topic:
A major effect of aprocitentan on albuminuria in patients with resistant hypertension
Schlaich Markus, Bakris George, Flack John, Gimona Alberto, Narkiewicz Krzysztof, Sassi-sayadi Mouna, Wang Jiguang, Weber Michael
A Quantitative Comparison of Varying Calcium Plaque Morphology Using High Resolution Micro-CT in Cadaveric Left Anterior Descending ArteriesPham Huynh Tien, Kim Alexis, Hurdle Kelsi, Beatty Brian, Onasoga Moyosore, Elci Gianna, Chong Lionel, Simha Pradyumna, Magnotto John, Mann Shahzabe, Husain Gazi, Fomin Jacob