Final ID: Su1143
Platelet Activation in Patients with Peripheral Artery Disease From 43 Sites: Assessment of Samples Transported by Overnight Mail
Abstract Body (Do not enter title and authors here): Background: Platelet activation plays a key role in peripheral artery disease (PAD), but the relevant platelet activation mechanism in chronic limb threatening ischemia remains unclear. Moreover, multi-center study of platelet activation remains difficult due to methodologic limitations. We report the feasibility of a novel method to measure platelet activation using samples sent via overnight mail. The primary objective was to report baseline markers of platelet activation from samples sent by overnight mail compared to an age-matched population recruited locally without known atherosclerosis.
Methods: Peripheral venous blood was drawn from 190 participants with severe peripheral artery disease at 43 sites in the BEST-CLI (Best Endovascular Versus Best Surgical Therapy in Patients with Chronic Limb Ischemia) Trial and shipped by Federal Express cold pack from study site locations within the continental United States to Vanderbilt University Medical Center. Fifty-six volunteers with and without diabetes were recruited locally as time and age-matched control subjects. Samples were collected using a platelet activation inhibitor. Thromboxane B2, dilysyl-malondialdehyde (MDA), p-selectin, WEDE 15, and SPAN 12 as markers of platelet activation were measured using liquid chromatography with tandem mass spectrometry and flow cytometry.
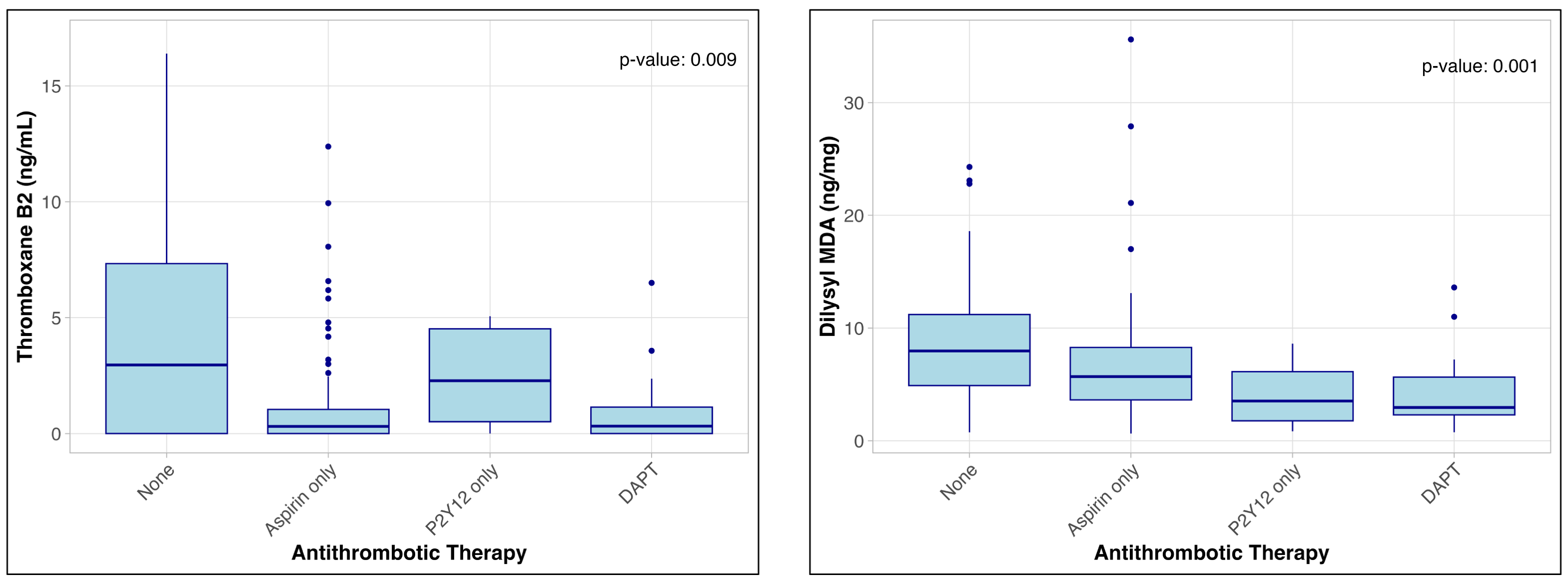
Results: Compared with control subjects, patients from the BEST-CLI trial were more likely to be male (72.6% vs 51.8%, p<0.01), have prior atherosclerotic cardiovascular disease (22.1% vs 0%, p<0.01), and be prescribed aspirin (69.4% vs 35.7%, p<0.01) or clopidogrel (20.0% vs 0%, p<0.01). Thromboxane B2 (0.5 vs 2.9 ng/mL, p<0.01) and dilysyl-MDA (4.7 vs 9.7 ng/mg, p<0.01) were significantly higher in BEST-CLI patients than control subjects while p-selectin (592.0 vs 420.5 MFI, p<0.01) was lower. Levels of thromboxane B2 (figure 1A) and dilysyl-MDA (figure 1B) varied significantly and as expected across the range of antithrombotic prescription (p<0.01).
Conclusions: Measurement of platelet activation after sample shipment on cold pack is feasible and yields reliable results that match expected clinical values.
Methods: Peripheral venous blood was drawn from 190 participants with severe peripheral artery disease at 43 sites in the BEST-CLI (Best Endovascular Versus Best Surgical Therapy in Patients with Chronic Limb Ischemia) Trial and shipped by Federal Express cold pack from study site locations within the continental United States to Vanderbilt University Medical Center. Fifty-six volunteers with and without diabetes were recruited locally as time and age-matched control subjects. Samples were collected using a platelet activation inhibitor. Thromboxane B2, dilysyl-malondialdehyde (MDA), p-selectin, WEDE 15, and SPAN 12 as markers of platelet activation were measured using liquid chromatography with tandem mass spectrometry and flow cytometry.
Results: Compared with control subjects, patients from the BEST-CLI trial were more likely to be male (72.6% vs 51.8%, p<0.01), have prior atherosclerotic cardiovascular disease (22.1% vs 0%, p<0.01), and be prescribed aspirin (69.4% vs 35.7%, p<0.01) or clopidogrel (20.0% vs 0%, p<0.01). Thromboxane B2 (0.5 vs 2.9 ng/mL, p<0.01) and dilysyl-MDA (4.7 vs 9.7 ng/mg, p<0.01) were significantly higher in BEST-CLI patients than control subjects while p-selectin (592.0 vs 420.5 MFI, p<0.01) was lower. Levels of thromboxane B2 (figure 1A) and dilysyl-MDA (figure 1B) varied significantly and as expected across the range of antithrombotic prescription (p<0.01).
Conclusions: Measurement of platelet activation after sample shipment on cold pack is feasible and yields reliable results that match expected clinical values.
More abstracts on this topic:
Acute Myocardial Infarction outcomes in patients on single anticoagulation versus combined anticoagulation and antiplatelet therapy
Rosario Manuel, Vasquez Moises, Garcia Gustavo, Wojtas Caroline, Vargas Ian, Behers Benjamin
Antithrombotic trends before and after publication of randomized clinical trials in cervical artery dissection: A secondary analysis of the STOP-CAD StudyPenckofer Mary, Salehi Omran Setareh, Seiffge David, Arnold Marcel, Marialuisa Zedde, Zubair Adeel, Marto Joao Pedro, Ghannam Malik, Engelter Stefan, Traenka Christopher, Mac Grory Brian, Shu Liqi, Kam Wayneho, Elnazeir Marwa, Romoli Michele, Saleh Velez Faddi, Siegler James, Strelecky Lukas, Yaghi Shadi, Henninger Nils, Muppa Jayachandra, Bakradze Ekaterina, Heldner Mirjam, Katheryna Antonenko