Final ID: MDP1396
Clinical outcomes of cardiac synchronization with or without an implantable cardioverter defibrillator based on pooled data from 5 clinical trials: a patient-level meta-analysis
Abstract Body (Do not enter title and authors here): Background: Cardiac resynchronization therapy (CRT) is a well-established therapy for patients with heart failure with reduced ejection fraction (HFrEF) and wide QRS. Whether CRT-defibrillators (CRT-D) reduce mortality more than CRT-pacemakers (CRT-P) remains controversial.
Aims: To compare the clinical outcomes of CRT-D vs CRT-P using data from 5 landmark CRT trials, both overall and stratified by etiology of cardiomyopathy (ischemic vs non-ischemic), sex (male vs female), age (≥ 70 y/o vs < 70 y/o), and QRS morphology (IVCD, LBBB, RBBB).
Methods: We performed a meta-analysis of patient level data from 5 prospective CRT trials (MIRACLE, REVERSE, RAFT, COMPANION and MADIT-CRT). Inclusion criteria were CRT-P vs CRT-D status (randomized comparison only in COMPANION), age ≥ 18 y/o and LVEF ≤ 35%. Exclusion criteria included secondary prevention ICD, QRS < 120ms, pacemaker upgrade, ventricular pacing indication, or missing data. Primary outcome was composite of time to heart failure hospitalization (HFH) or all-cause death. Secondary outcomes were time to HFH and death. Outcomes were analyzed using a frequentist Cox Proportional Hazards mixed effects model adjusted for 17 variables.
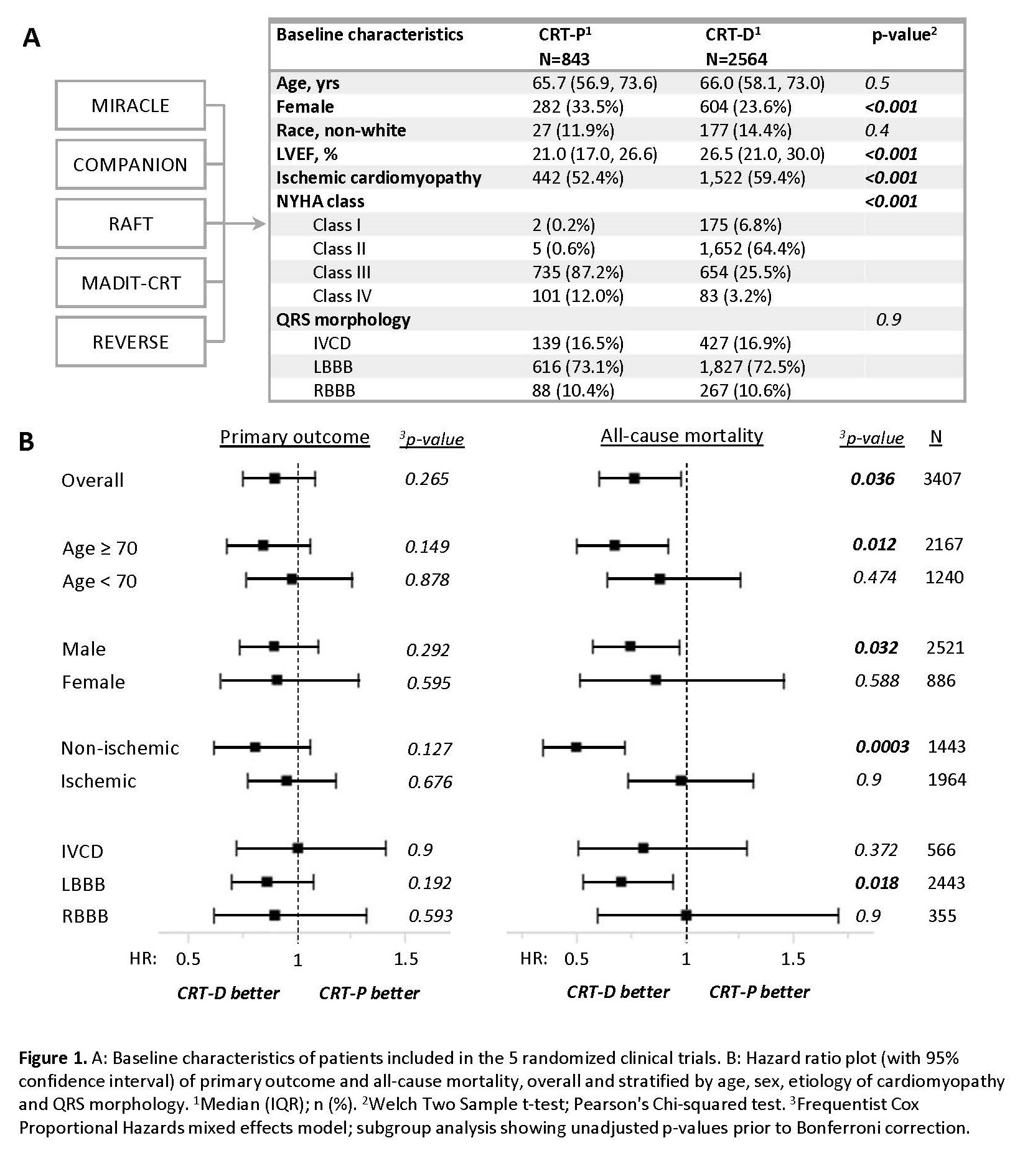
Results: A total of 3407 patients met inclusion criteria. Relative to patients with CRT-P (n=843), those with CRT-D (n=2564) were of similar age (66 y/o, p=0.5), less often female (24% vs 34%, p<0.001), and more often had ischemic cardiomyopathy (59.4% vs 52.4%, p<0.001), Fig 1A. Primary outcome was similar across groups (HR 0.902 [0.752, 1.081], p=0.26), but all-cause mortality was lower with CRT-D vs CRT-P (HR 0.77 [0.603, 0.983], p=0.036), Fig 1B. Interaction analyses suggested lower all-cause mortality with CRT-D vs CRT-P in patients with non-ischemic cardiomyopathy (HR 0.502 [0.346, 0.726], p=0.0003) and patients age ≥70 y/o (HR 0.679 [0.502, 0.919], p=0.012), with significance preserved after Bonferroni correction (Fig 1B).
Conclusion: In patients receiving CRT for HFrEF, those with CRT-D had lower all-cause mortality than patients with CRT-P, driven mainly by a lower mortality with CRT-D in older patients and those with non-ischemic cardiomyopathy. No significant interactions were noted between ICD & sex or ICD & QRS morphology.
Aims: To compare the clinical outcomes of CRT-D vs CRT-P using data from 5 landmark CRT trials, both overall and stratified by etiology of cardiomyopathy (ischemic vs non-ischemic), sex (male vs female), age (≥ 70 y/o vs < 70 y/o), and QRS morphology (IVCD, LBBB, RBBB).
Methods: We performed a meta-analysis of patient level data from 5 prospective CRT trials (MIRACLE, REVERSE, RAFT, COMPANION and MADIT-CRT). Inclusion criteria were CRT-P vs CRT-D status (randomized comparison only in COMPANION), age ≥ 18 y/o and LVEF ≤ 35%. Exclusion criteria included secondary prevention ICD, QRS < 120ms, pacemaker upgrade, ventricular pacing indication, or missing data. Primary outcome was composite of time to heart failure hospitalization (HFH) or all-cause death. Secondary outcomes were time to HFH and death. Outcomes were analyzed using a frequentist Cox Proportional Hazards mixed effects model adjusted for 17 variables.
Results: A total of 3407 patients met inclusion criteria. Relative to patients with CRT-P (n=843), those with CRT-D (n=2564) were of similar age (66 y/o, p=0.5), less often female (24% vs 34%, p<0.001), and more often had ischemic cardiomyopathy (59.4% vs 52.4%, p<0.001), Fig 1A. Primary outcome was similar across groups (HR 0.902 [0.752, 1.081], p=0.26), but all-cause mortality was lower with CRT-D vs CRT-P (HR 0.77 [0.603, 0.983], p=0.036), Fig 1B. Interaction analyses suggested lower all-cause mortality with CRT-D vs CRT-P in patients with non-ischemic cardiomyopathy (HR 0.502 [0.346, 0.726], p=0.0003) and patients age ≥70 y/o (HR 0.679 [0.502, 0.919], p=0.012), with significance preserved after Bonferroni correction (Fig 1B).
Conclusion: In patients receiving CRT for HFrEF, those with CRT-D had lower all-cause mortality than patients with CRT-P, driven mainly by a lower mortality with CRT-D in older patients and those with non-ischemic cardiomyopathy. No significant interactions were noted between ICD & sex or ICD & QRS morphology.
More abstracts on this topic:
A Bridge from Sweet to Sour: A Case of Recurrent Myocardial Stunning in Diabetic Ketoacidosis
Satish Vikyath, Pargaonkar Sumant, Slipczuk Leandro, Schenone Aldo, Maliha Maisha, Chi Kuan Yu, Sunil Kumar Sriram, Borkowski Pawel, Vyas Rhea, Rodriguez Szaszdi David Jose Javier, Kharawala Amrin, Seo Jiyoung
A Growing Burden of Electronic Medical Record Messages in ACHD CareDailey Schwartz Andrew, Alegria Jorge