Final ID: MDP1620
Utility of Coronary Artery Calcium Scoring in Low-Risk Patients: the Multi-Ethnic Study of Atherosclerosis (MESA)
Abstract Body (Do not enter title and authors here): Introduction: Coronary artery calcium (CAC) scoring is a powerful tool for atherosclerotic cardiovascular disease (ASCVD) risk assessment. Guidelines recommend consideration of CAC scoring in intermediate risk patients, but it’s utility in low and borderline risk patients is less clear.
Methods: We used data from 2,894 participants from MESA with low (<5%) or borderline (5-<7.5%) 10-year ASCVD risk defined by the pooled cohort equations (PCE). We evaluated the association between CAC score and coronary heart disease (CHD) and ASCVD risk using multivariable adjusted Cox proportional hazard models. We also evaluated if the addition of CAC to the pooled cohort equations (PCE) improved risk prediction using Harrell’s C-index and net reclassification improvement (NRI).
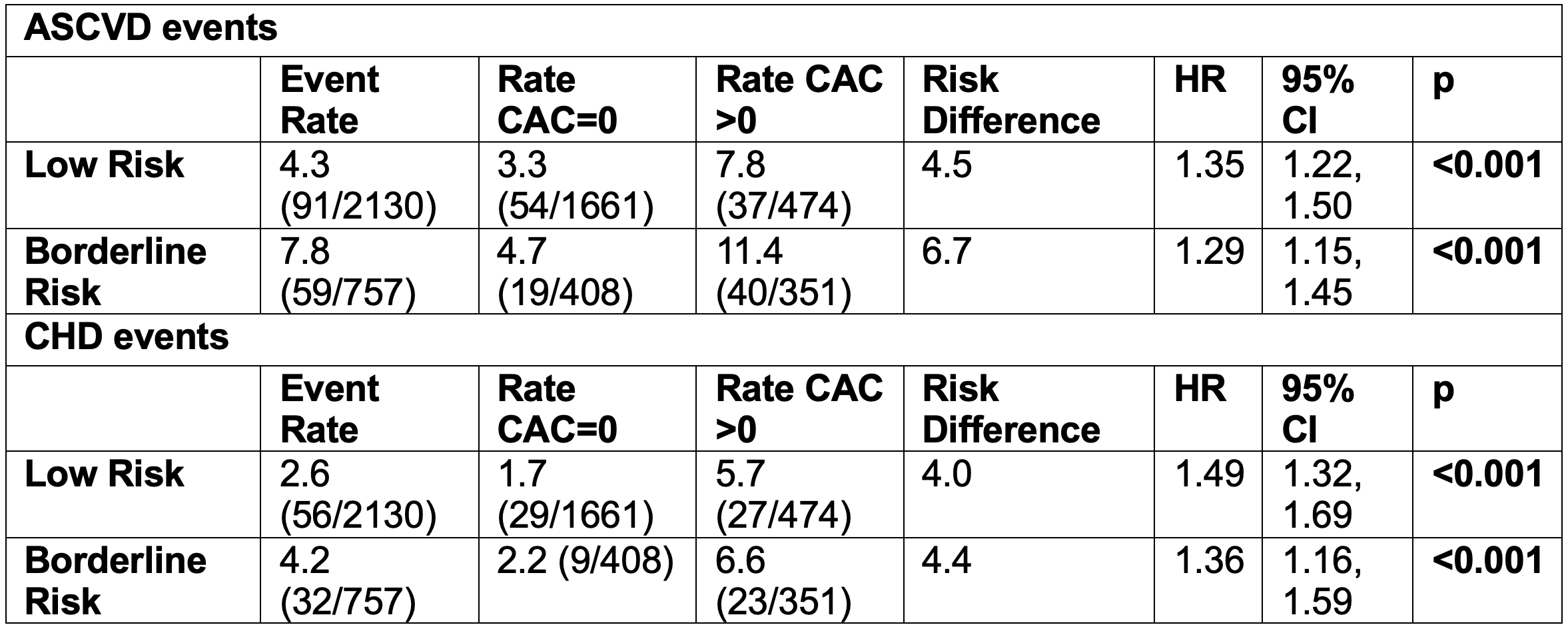
Results/Data: Mean age was 54.1 ± 5.8 years with 66.9% women. Among low-risk individuals, the ASCVD event rate was 3.3% with CAC=0, and 7.8% with CAC>0, and 4.7% vs. 11.4% among borderline-risk individuals. Natural-log transformed CAC score was associated with increased ASCVD risk in low-risk (HR 1.35, 95% CI 1.22-1.50) and borderline-risk (HR 1.29, 95% CI 1.15-1.45) individuals. Similar results were seen for CHD with higher HRs (Table 1). The addition of CAC to the PCE improved the C-index (SE) among low/borderline risk individuals from 0.620 (0.023) to 0.671 (0.024). Continuous NRI was also significant with the addition of CAC to the PCE in low/borderline risk (0.422, 95% CI 0.248-0.611). Greater improvement was seen for CHD (Table 2).
Conclusions: The presence of CAC in low and borderline risk individuals is associated with increased ASCVD and CHD risk. The addition of CAC scoring to the PCE improved risk prediction in low and borderline risk individuals, suggesting potential clinical utility in this population.
Methods: We used data from 2,894 participants from MESA with low (<5%) or borderline (5-<7.5%) 10-year ASCVD risk defined by the pooled cohort equations (PCE). We evaluated the association between CAC score and coronary heart disease (CHD) and ASCVD risk using multivariable adjusted Cox proportional hazard models. We also evaluated if the addition of CAC to the pooled cohort equations (PCE) improved risk prediction using Harrell’s C-index and net reclassification improvement (NRI).
Results/Data: Mean age was 54.1 ± 5.8 years with 66.9% women. Among low-risk individuals, the ASCVD event rate was 3.3% with CAC=0, and 7.8% with CAC>0, and 4.7% vs. 11.4% among borderline-risk individuals. Natural-log transformed CAC score was associated with increased ASCVD risk in low-risk (HR 1.35, 95% CI 1.22-1.50) and borderline-risk (HR 1.29, 95% CI 1.15-1.45) individuals. Similar results were seen for CHD with higher HRs (Table 1). The addition of CAC to the PCE improved the C-index (SE) among low/borderline risk individuals from 0.620 (0.023) to 0.671 (0.024). Continuous NRI was also significant with the addition of CAC to the PCE in low/borderline risk (0.422, 95% CI 0.248-0.611). Greater improvement was seen for CHD (Table 2).
Conclusions: The presence of CAC in low and borderline risk individuals is associated with increased ASCVD and CHD risk. The addition of CAC scoring to the PCE improved risk prediction in low and borderline risk individuals, suggesting potential clinical utility in this population.
More abstracts on this topic:
A Scoping Review Exploring Cardiovascular Risk and Health Metrics and Cancer Prediction
Kim Ji-eun, Henriquez Santos Gretell, Kumar Sant, Livinski Alicia, Vo Jacqueline, Joo Jungnam, Shearer Joe, Hashemian Maryam, Roger Veronique
Agbaje’s Waist-to-Height Ratio Estimated Fat Mass Pediatric Cutoff Predicts Elevated Blood Pressure Risk in Multi-racial US Children and AdolescentsCorsi Douglas, Agbaje Andrew